Publications
2026
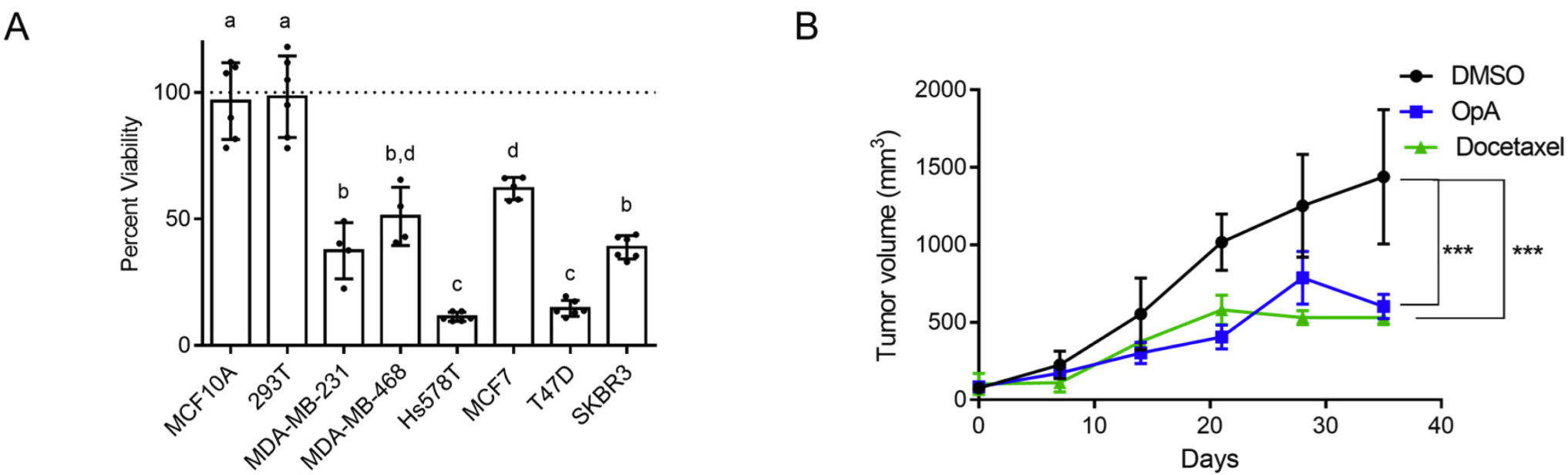
183. Gasdermin D Cleavage and Cytokine Release, Indicative of Pyroptotic Cell Death, Induced by Ophiobolin A in Breast Cancer Cell Lines Ranganathan, S.; Ojo, T.; Subramanian, A.; Tobin, J.; Kornienko, A.; Boari, A.; Evidente, A.; Benton, M. L.; Romo, D.; Taube, J. H. Int. J. Mol. Sci. 2026, 27, 2, 618.
2025
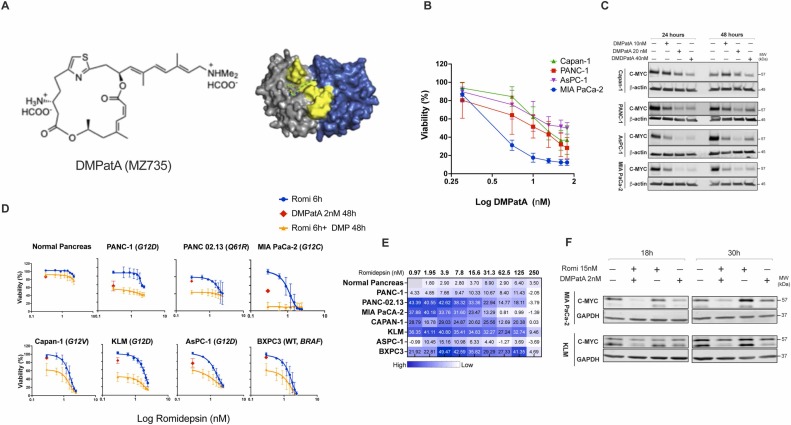
182. Combined HDAC and eIF4A inhibition: A novel epigenetic therapy for pancreatic ductal adenocarcinoma Safari, M.; Scotto, L.; Basseville, A.; Litman, T.; Xue, H.; Petrukhin, L.; Zhou, P.; Remotti, H. E.; Ku, A.; Morales, D. V.; Damoci, C.; Zhu, M.; Maddipati, R.; Hull, K. G.; Robey, R. W.; Olive, K. P.; Fojo, T.; Romo, D.; Bates, S. E. Drug Resistance Updates. 2025, Preproof.
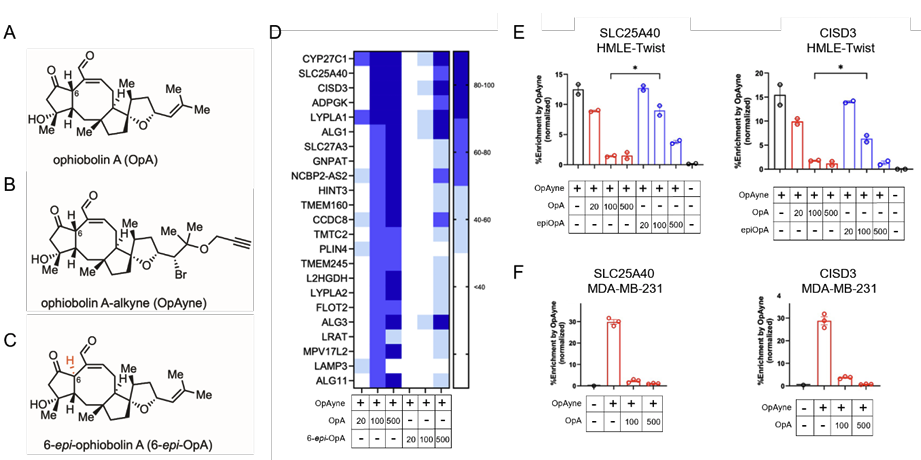
181. Ophiobolin A selectively alters mitochondria, metabolism and redox biology in breast cancer and mammary epithelial cells which have undergone epithelial to mesenchymal transition Parker, H. N.; Tao, Y.; Tobin, J.; Haberman, K. L.; Davis, S.; York, E.; Martinez, A.; Matsumoto, N.; Aroujo, J.; Park, J. H.; Zechmann, B.; Kaipparettu, B. A.; Boari, A.; Sayes, C. M.; Evidente, A.; Kornienko, A.; Cravatt, B.; Romo, D.; Taube, J. H. bioRxiv preprint. 2025.
180. Derivatization of ophiobolin A and cytotoxicity toward breast and glioblastoma cancer stem cells: Varying the ketone and unsaturated aldehyde moieties Aroujo, J.; Parker, H.; Boari, A.; Mason, E.; Otakpor, M. U.; Betancourt, T.; Kornienko, A.; Ciavatta, M.; Carbone, M.; Evidente, A.; Taube, J. H.; Romo, D. Bioorg. Med. Chem. Lett. 2025, 120, 9, 130112.

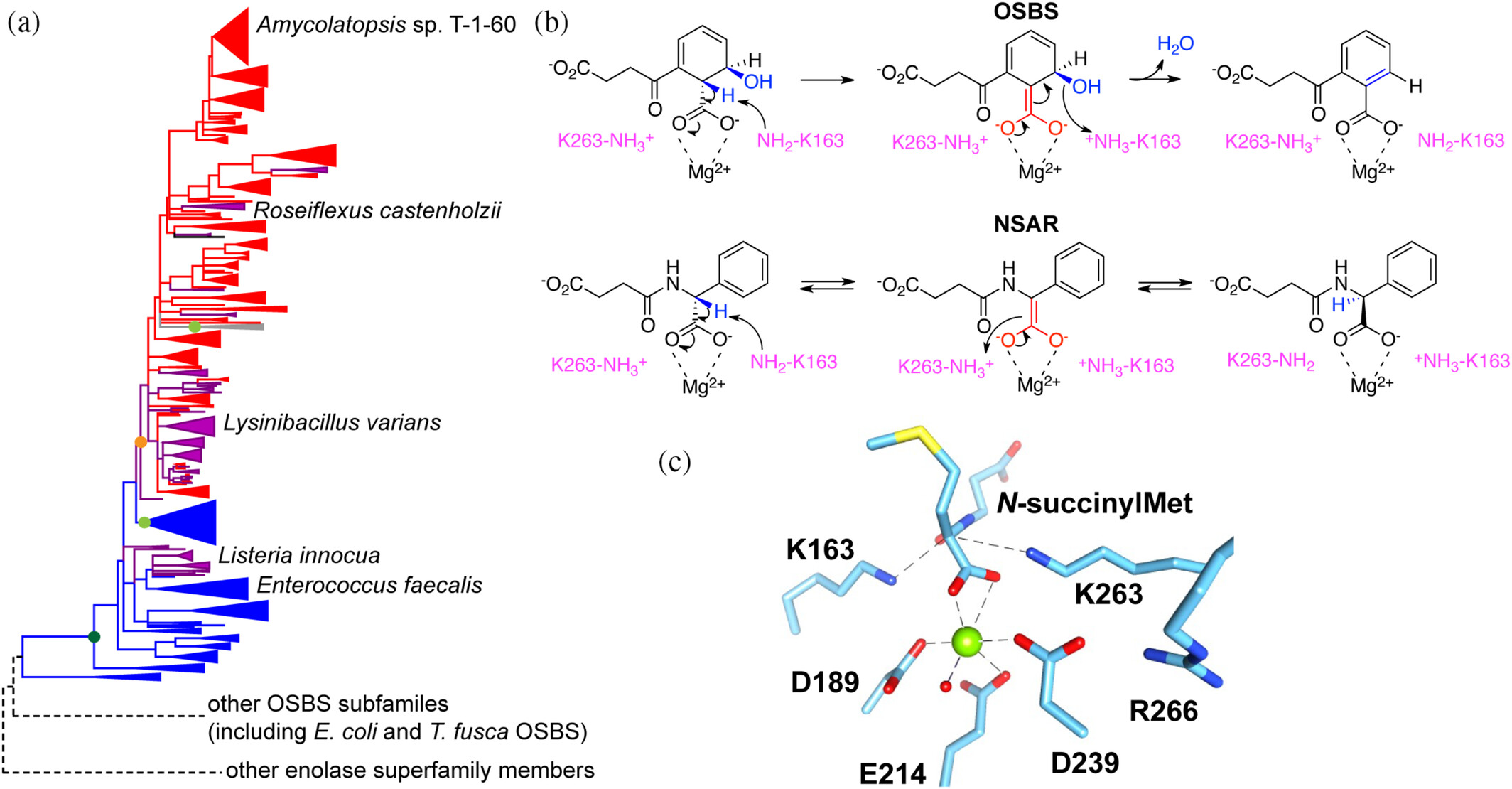
179. Intramolecular epistasis correlates with divergence of specificity in promiscuous and bifunctional NSAR/OSBS enzymes Truong, D. P.; Dharmatti, R.; Suriadinata, D.; Huddleston, J.; Skouby, R.; Addo, G. O.; Delpe Acharige, A.; Bayanaya, R. S.; Davila, C.; Fults, S. C.; Raushel, F. M.; Hull, K. G.; Romo, D.; Glasner, M. E. Protein Science. 2025, e70113.
2024
178. TRPM7 in neurodevelopment and therapeutic prospects for neurodegenerative disease Luo, Z.; Zhang, X.; Fleig, A.; Romo, D.; Hull, K. G.; Horgen, D. F.; Sun, H.; Feng, Z. Cell Calcium 2024, 120, 102886.

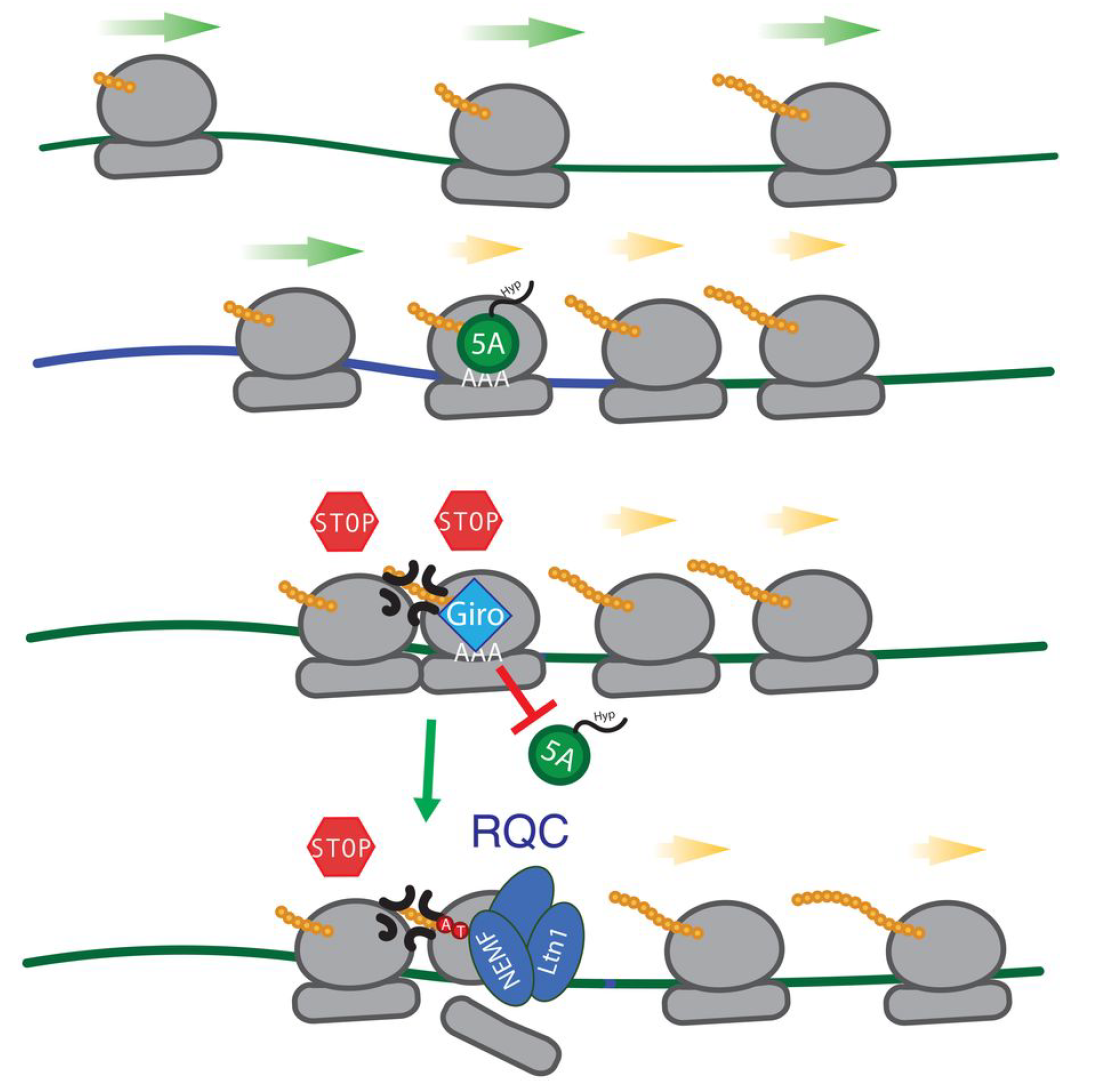
177. Girolline is a sequence-context specific modulator of eIF5A activity Dang, Y.; Arata, M.; Shichino, Y.; Al Mourabit, A.; Romo, D.; Liu, J.; Iwasaki, S.; Yoshida, M.; Schneider-Poetsch, T. . Nat. Comm. 2024, 120, 102886.
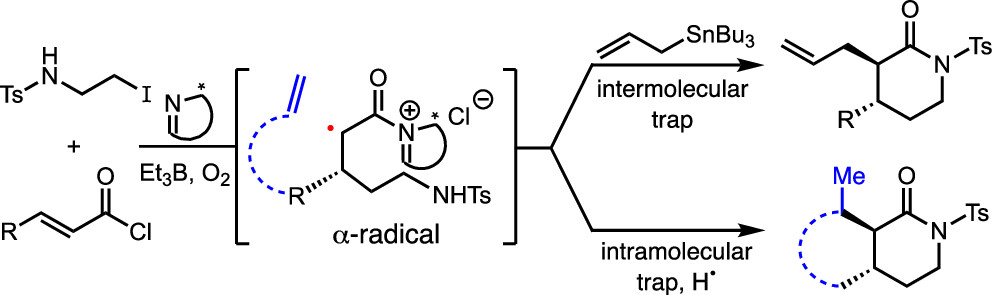
176. Radical Reactions with Chiral Acylammonium Salts: Synthesis of Functionalized δ-Lactams through Giese-Initiated Organocascades Sutter, P. J.; Walker, B.; Kasper, K.; Kojasoy, V.; Tantillo, D. J.; Romo, D. J. Org. Chem. 2024, 89, 19, 14209-14216.
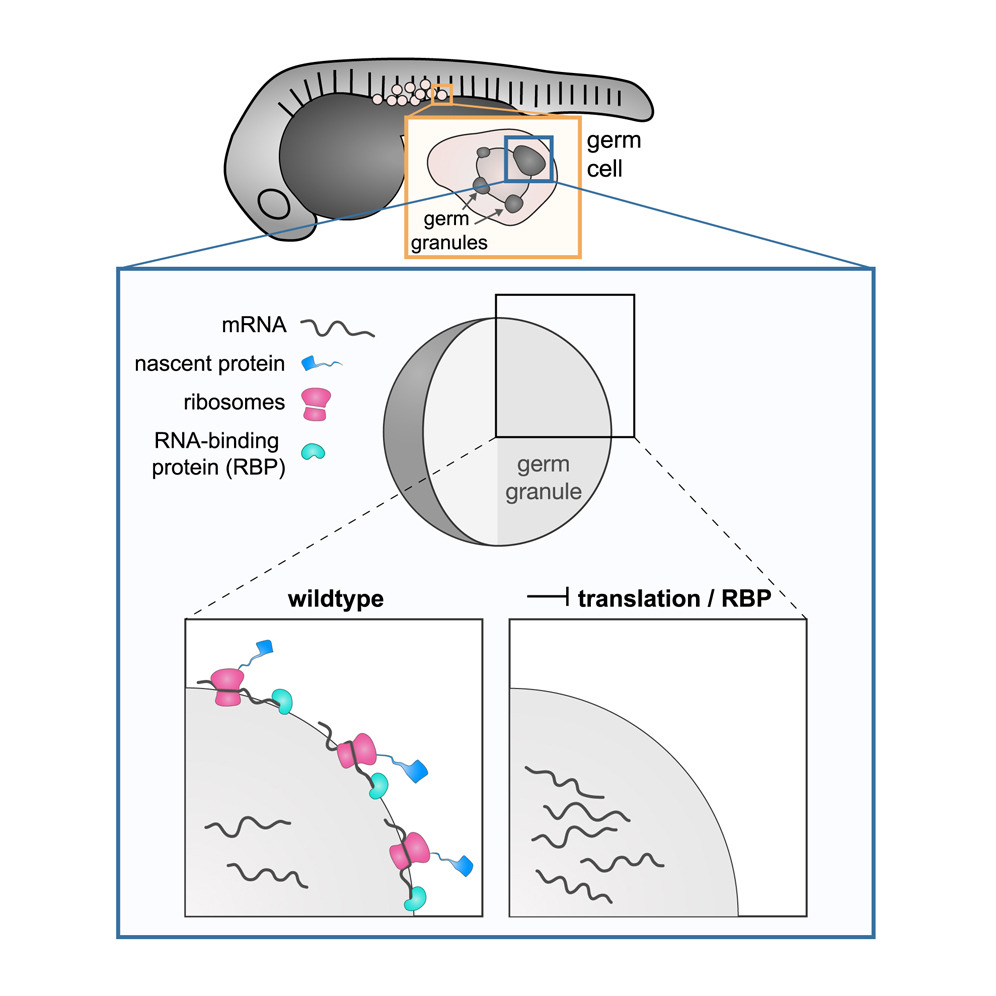
175. Spatial organization and function of RNA molecules within phase-separated condensates in zebrafish are controlled by Dnd1 Westerech, K. J.; Tarbashevich, K.; Schick, J.; Gupta, A.; Zhu, M.; Hull, K.; Romo, D.; Zeuschner, D.; Goudarzi, M.; Gross-Thebing, T.; Raz, E. Dev. Cell 2023, 58, 17, 1578-1592.e5
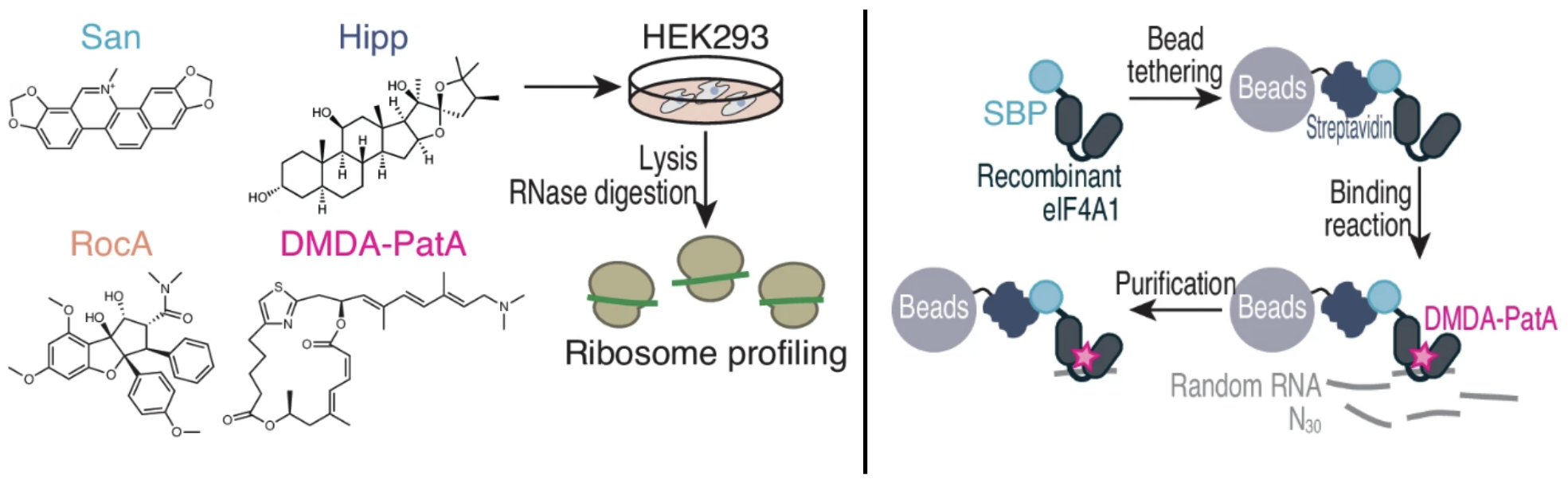
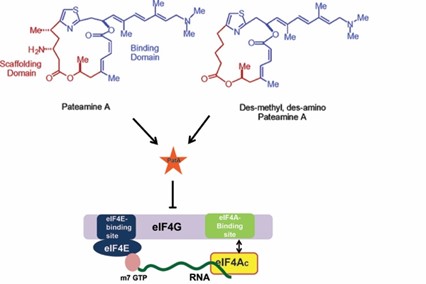
174. DMDA-PatA mediates RNA sequence-selective translation repression by anchoring eIF4A and DDX3 to GNG motifs Saito, H.; Handa, Y.; Chen, M.; Schneider-Poetsch, T.; Shichino, Y.; Takahashi, M.; Romo, D.; Yoshida, M.; Furstner, A.; Ito, T.; Fukluzawa, K.; Iwasaki, S. Nat. Comm. 2024, 7418
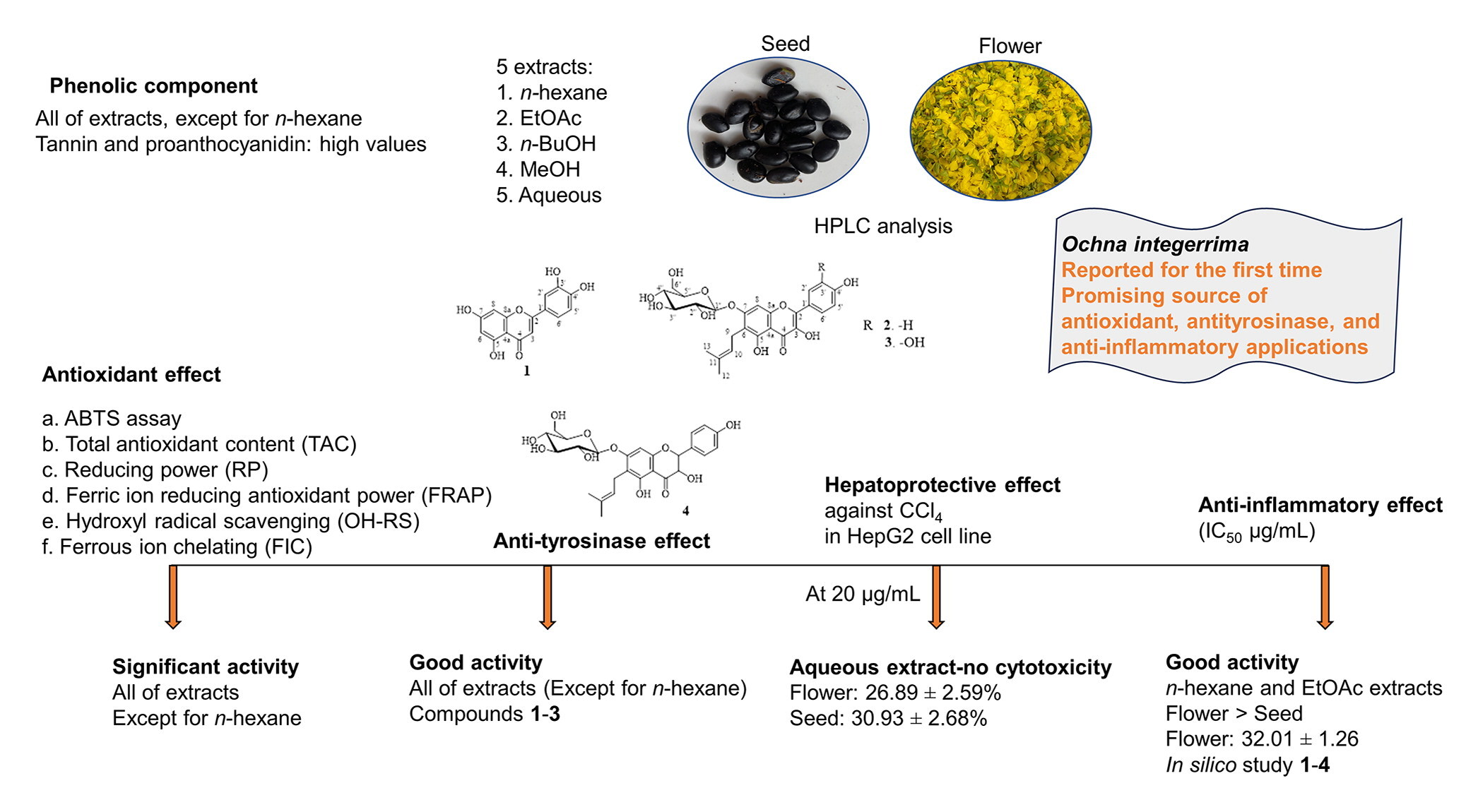
173. Antioxidant, anti-tyrosinase, hepatoprotective, and anti-inflammatory potential in flowers and seeds of Ochna integerrima (Lour.) Merr Nguyen, H. M.; Nguyen, K. P.; Le, A. T. P.; Nguyen, N. H. T.; Vu-Huynh, L.; K.; Le, C. K. T.; Acharige, A. D.; Hull, K.; Romo, D. Nat. Prod. Res. 2024, 1-11.
172. Dopamine-Derived Guanidine Alkaloids from a Didemnidae Tunicate: Isolation, Synthesis, and Biological Activities Sakai, R.; Matsumara, K.; Uchimasu, H.; Miyako, K.; Taniguchi, T.; Kovvuri, R. R.; Acharige, A. D.; Hull, K. G.; Romo, D.; Thaveepornkul, L.; Chimnaronk, S.; Miyamoto, H.; Takada, A.; Watari, H.; Fujita, M. J.; Sakaue, J. J. Org. Chem. 2024, 89, 9, 5977-5987.

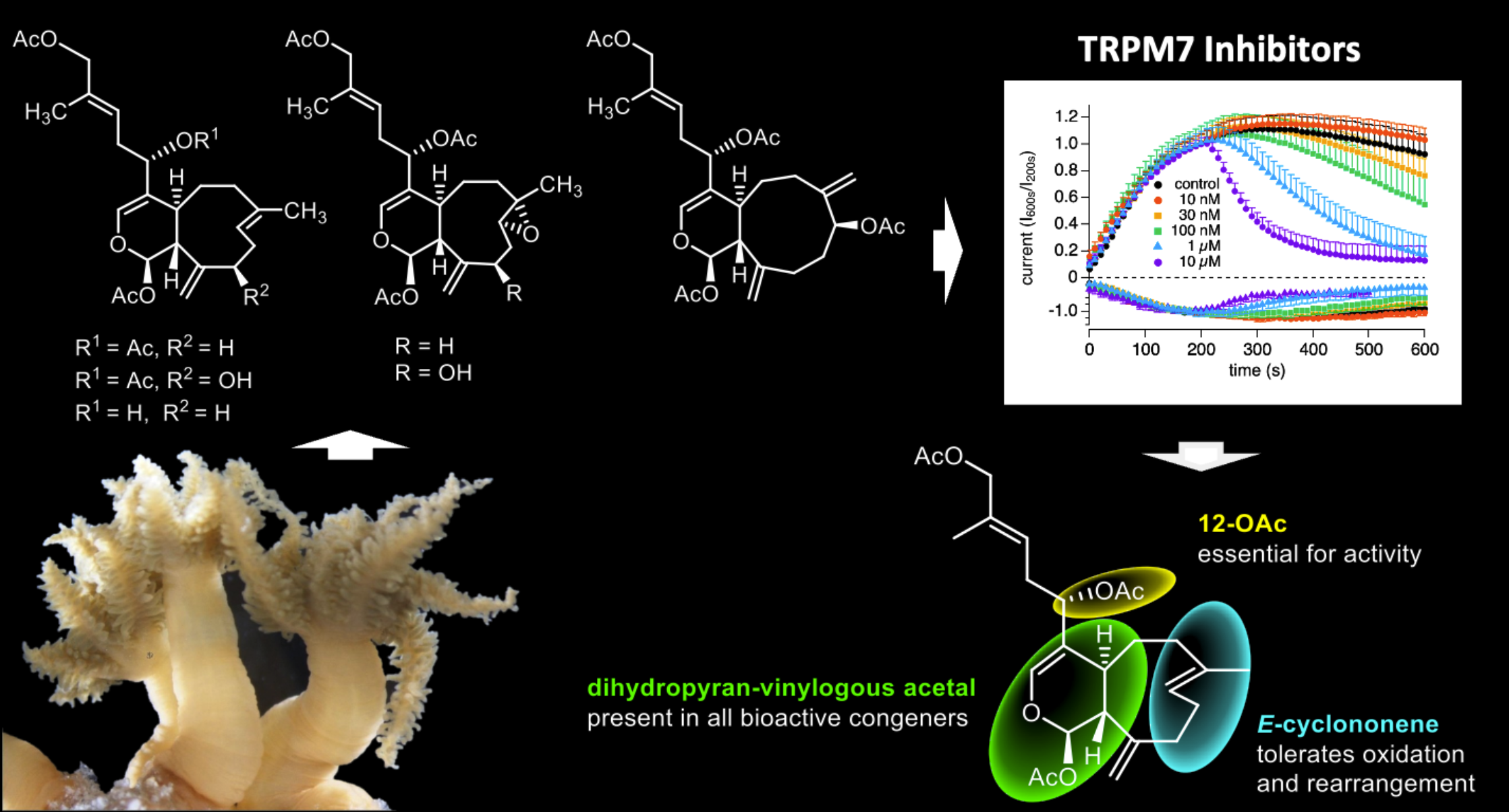
171. Transient Receptor Potential Melastatin 7 (TRPM7) Ion Channel Inhibitors: Preliminary SAR and Conformational Studies of Xenicane Diterpenoids from the Hawaiian Soft Coral Sarcothelia edmondsoni Yao, G.; Parris, M.; Kuo, W. C.; Porzgen, P.; Castillo, B.; Mason, E. S.; Chinchilla, A.;/ Huang, J.; Suzuki, S.; Ross, R.; Akana, E.; Vander Schuit, S.; Miller, S. P.; RPenner, R.; Sun, H.; Feng, Z.; Hull, K. G.; Romo, D. Fleig, A.; Horgen, D. F. J. Nat. Prod. 2024,
2023
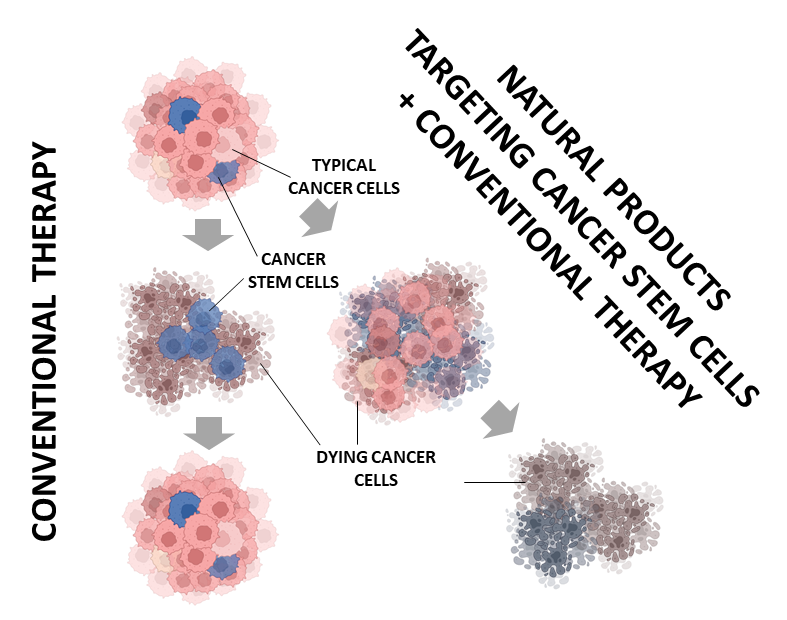
170. Therapeutic Vulnerabilities of Cancer Stem Cells and Effects of Natural Products Reisenauer, K.N.; Aroujo, J.; Tao, Y.; Ranganathan, S.; Romo, D.; Taube, J.H. Nat. Prod. Rep. 2023, Advance Article
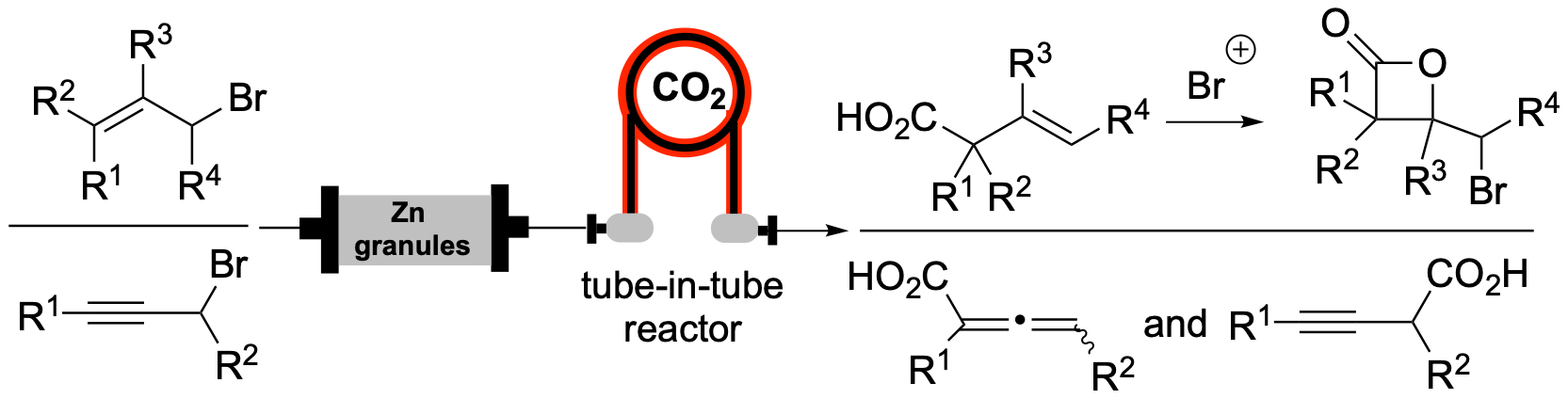
169. Zinc-mediated Carboxylations of Allylic and Propargylic Halides in Flow: Synthesis of β-Lactones via Subsequent Bromolactonization Sutter, P.J.; Kang, G.; Vellalath, S.; Romo, D. RSC Advances 2023, 3468 – 3473.
2022
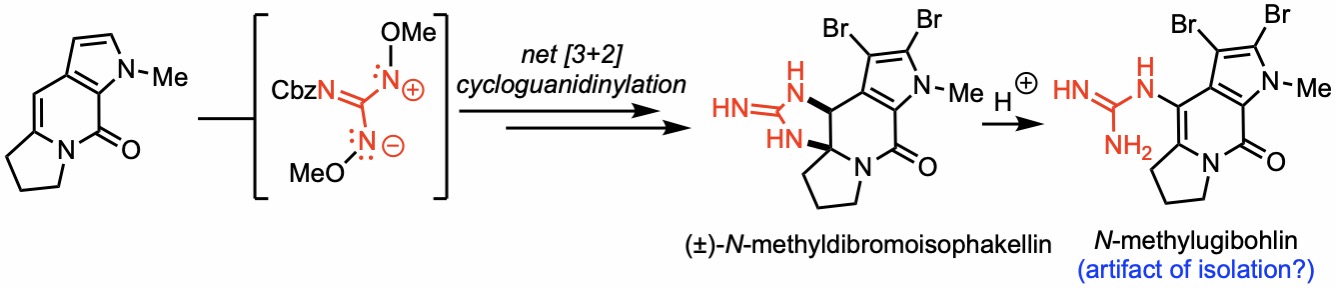
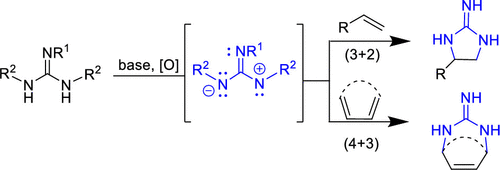
168. Total Synthesis of (±)-N-Methyldibromoisophakellin and N-Methylugibohlin via Net [3+2] Cycloguanidinylations Employing 2-Amido-1,3-Diamino-Allyl Cations Matsumoto, N.; Hwang, T.; Romo, D. Tetrahedron Lett. 2022, 115, 154304.
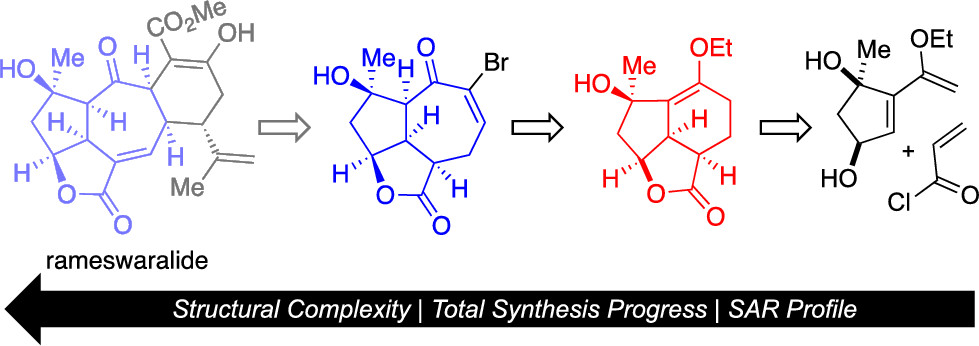
167. Total Synthesis of Rameswaralide Utilizing a Pharmacophore-Directed Retrosynthetic Strategy Truax, N.J.; Ayinde, S.; Liu, J.O.; Romo, D. J. Am. Chem. Soc. 2022, 144, 18575-18585.
[Featured in Synfacts; Designated Synfact of the Month]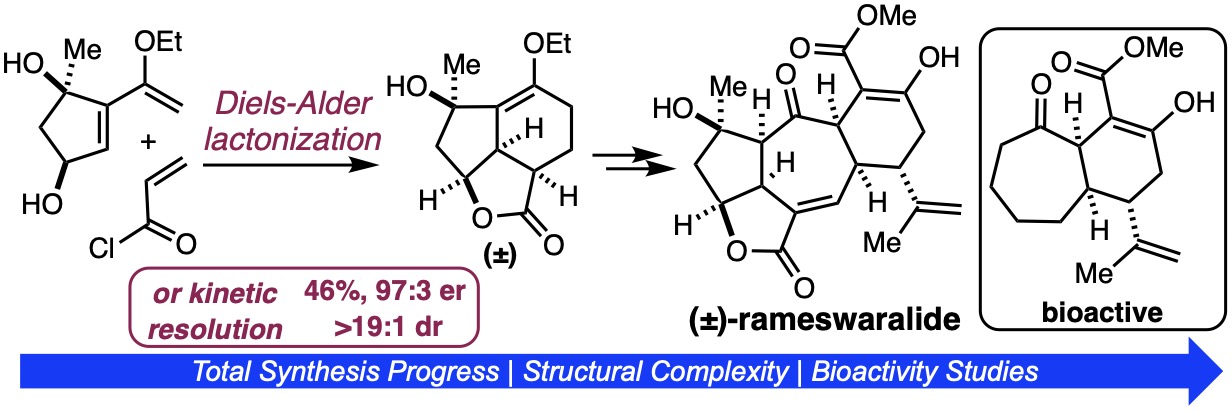
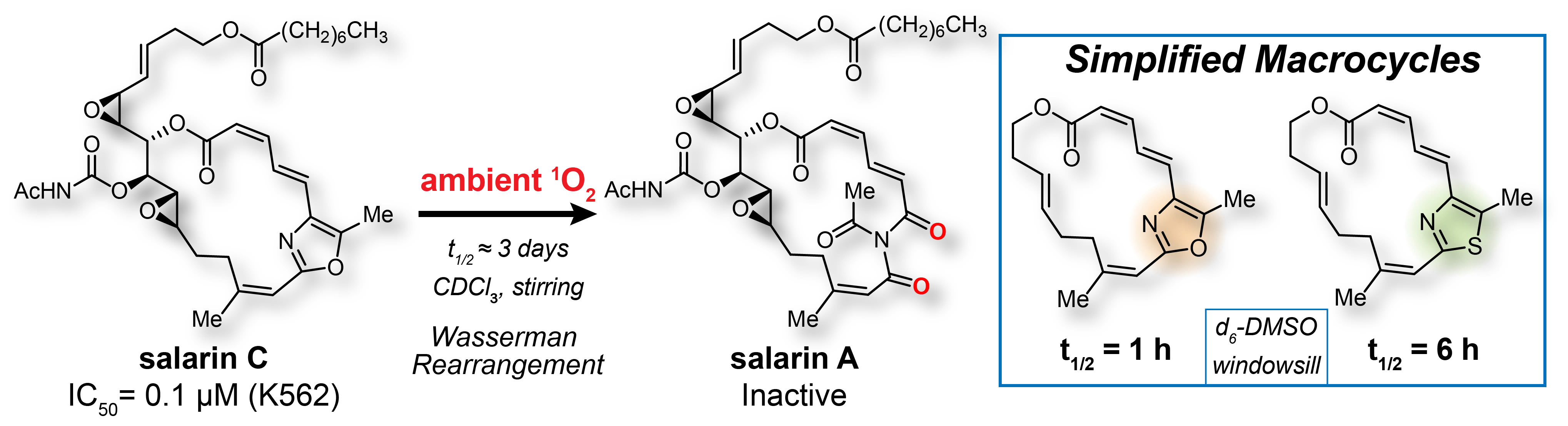
166. Synthesis and Stability Studies of a Simplified, Thiazole-Containing Macrocycle of the Anticancer Agent Salarin C Woods, J.E.; Jourdain, R.M.; Romo, D. Org. Lett. 2022, 24, 6891-6896
165. Enzymatic Assembly of the Salinosporamide γ-lactam-β-lactone Anticancer Warhead Bauman, K.D.; Shende, V. V.; Chen, P. Y-T., Trivella, D. B. B.; Gulder, T. A. M.; Vellalath, S.; Romo, D.; Moore, B. S. Nat. Chem. Biol. 2022, 18, 538-546
[Featured in C&EN]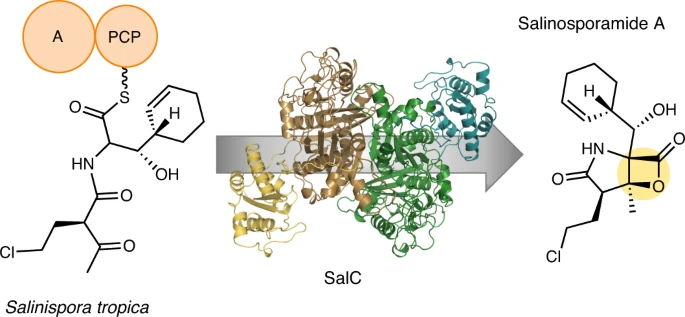
2021
164. Multi-component, Enantioselective Synthesis of Tetrahydropyridazinones Employing Chiral, α,ß-Unsaturated Acylammonium Salts Kiledal, S. A.; Jourdain, R.; Vellalath, S.; Romo, D. Org. Lett. 2021, 23, 6622-6627
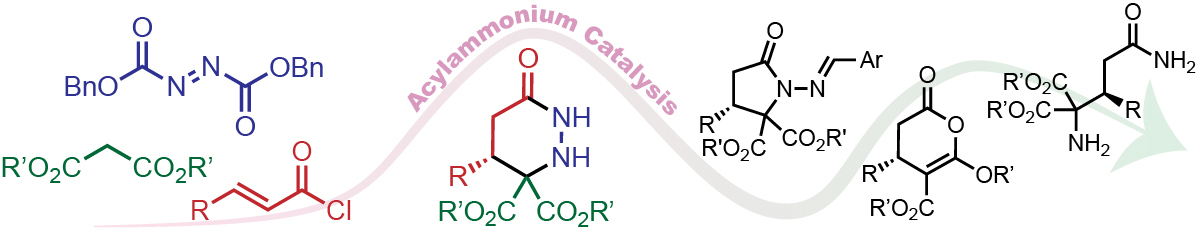
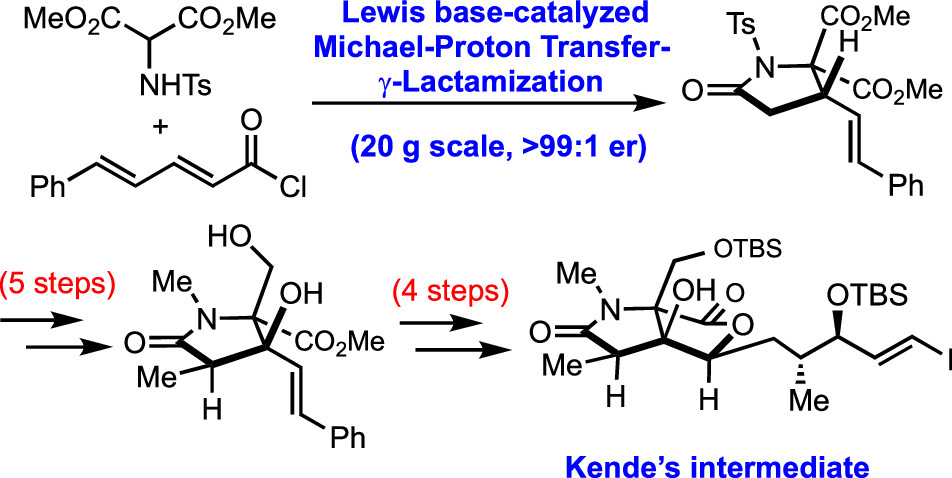
163. Enantioselective Michael-Proton Transfer-Lactamization for Pyroglutamic Acid Derivatives: Synthesis of Dimethyl-(S,E)-5-oxo-3-styryl-1-tosylpyrrolidine-2,2-dicarboxylate Chaheine, C. M.; Song, C. J.; Gladen, P. T.; Romo, D. Org. Synth. 2021, 98, 194-226
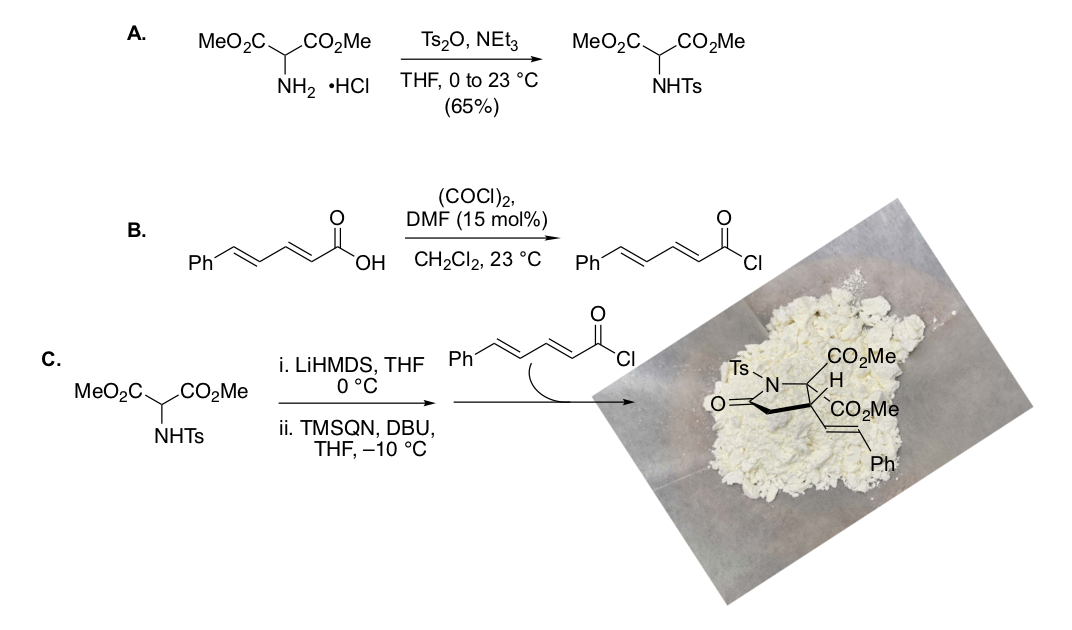
162. Synthesis of Agelastatin A and Derivatives Premised on a Hidden Symmetry Element Leading to Analogs Displaying Anticancer Activity Xue, H.; Svatek, H.; Bertonha, A. F.; Reisenauer, K.; Robinson, J., Kim, M.; Ingros, A.; Ho, M.; Taube, J.; Romo, D. Tetrahedron 2021, 94, 132340-132347
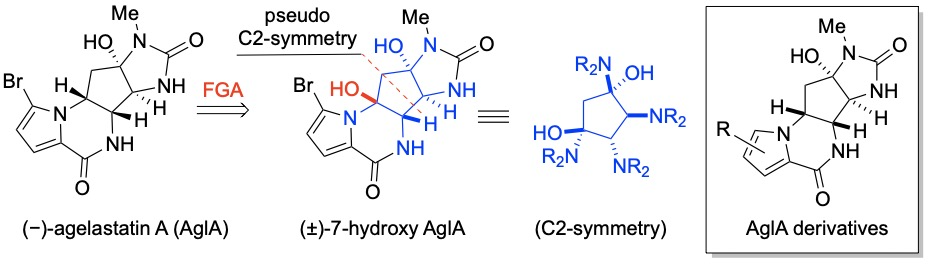
161. Epithelial-Mesenchymal Transition Sensitizes Breast Cancer Cells to Cell Death via the Fungus-Derived Sesterterpenoid Ophiobolin A Reisenauer, K. N.; Tao, Y.; Das, P.; Song, S.; Svatek, H.; Patel, S. D.; Mikhail, S.; Ingros, A.; Sheesley, P.; Masi, M.; Boari, A.; Evidente, A.; Kornienko, A.; Romo, D.; Taube, J. Scientific Reports 2021, 11, 10652-10662
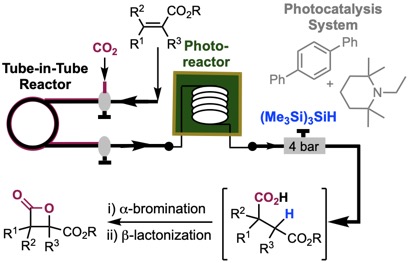
160. Photo-catalyzed, ß-Selective Hydrocarboxylation of α,ß-Unsaturated Esters with CO2 Under Flow for ß-Lactone Synthesis Kang, G.; Romo, D. ACS Catalysis, 2021, 11, 1309-1315.
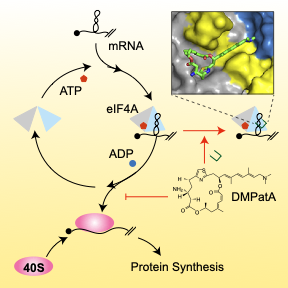
159. Functional mimicry revealed by the crystal structure of an eIF4A:RNA complex bound to the interfacial inhibitor, desmethyl pateamine A Nainen, S.K.; Liang, J.; Hull, K.; Cencic, R.; Zhu, M.; Northcote, P.; Teesdale-Spittle, P; Romo, D.; Nagar, B.; Pelletier, J. Cell Chem. Bio. 2021, 28, 1-10.
2020
158. Necator americanus Ancylostoma Secreted Protein-2 (Na-ASP-2) Binds an Ascaroside (ascr#3) in Its Fatty Acid Binding Site Otab O. E.; Darwiche, R.; Truax, N. J.; Schneiter, R.; Hull, K. G.; Romo, D.; Asojo, O. A. Front. Chem. 2020, 8, 608296.
157. Enantioselective, Organocatalytic Strategy for the Oxazolomycin Core: Formal Synthesis of (+)-Neooxazolomycin Chaheine, C. M.; Gladen, P. T.; Abbasov, M. E.; Romo, D. Org. Lett. 2020, 22, 9282-9286.
156. Editorial: Synthetic strategies for mining the information-rich content of natural products for biology and medicine Romo, D.; Vanderwal, C. D. Nat. Prod. Rep. 2020, 37, 1393-1394.
.png)
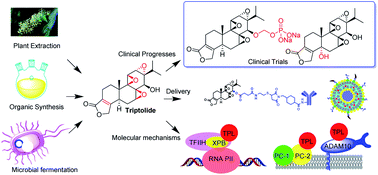
155. Triptolide: reflections on two decades of research and prospects for the future Tong, L.; Zhao, Q.; Datanb, E.; Linac, G.-Q.; Minnd, I.; Pomperd, M. G.; Yu, B.; Romo, D.; He, Q.-L.; Liu, J. O. Nat. Prod. Rep. 2020, Advance article.
154. Waixenicin A, a marine-derived TRPM7 inhibitor: a promising CNS drug lead Sun, H.-S.; Horgen, F. D.; Romo, D; Hull, K. G.; Kiledal, S. A.; Fleig, A.; Feng, Z.-P. Acta Pharmacol. Sin. 2020, 41, 1519–1524.
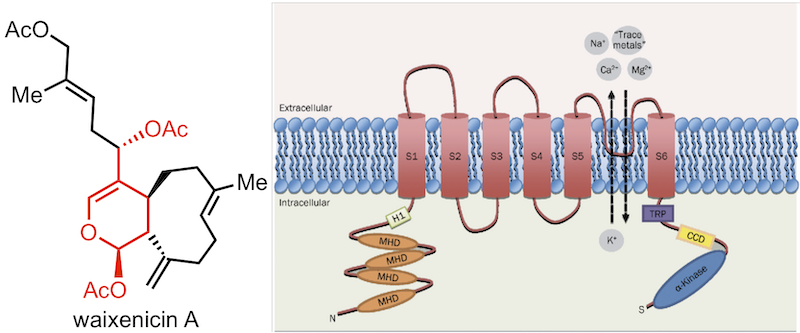
153. Bridging the gap between natural product synthesis and drug discovery Truax, N. J.; Romo, D. Nat. Prod. Rep. 2020, 37, 1436-1453.
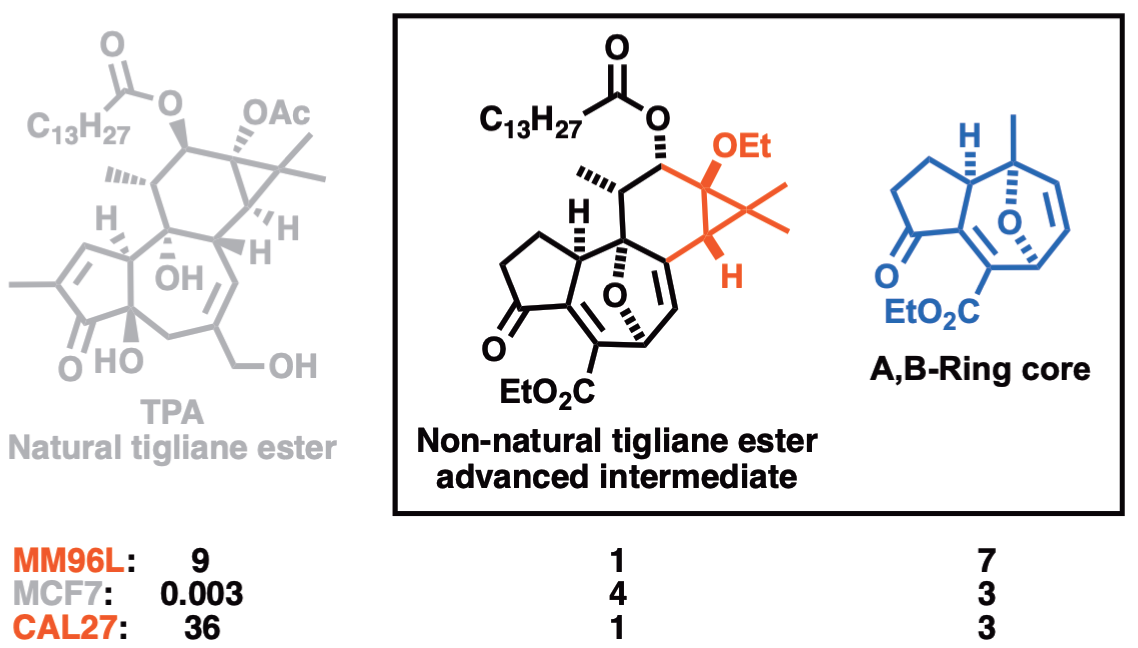
152. Synthetic Tigliane Intermediates Engage Thiols to Induce Potent Cell Line Selective Anti-Cancer Activity Chow, S.; Krainz, T.; Bettencourt, C.J.; Broit, N.; Ferguson, B.; Zhu, M.; Hull, K.G.; Pierens, G.K.; Bernhardt, P.V.; Parsons, P.G.; Romo, D.; Boyle, G.M.; Williams, C.M. Eur. J. Org. Chem. 2020, 26, 13372-13377.
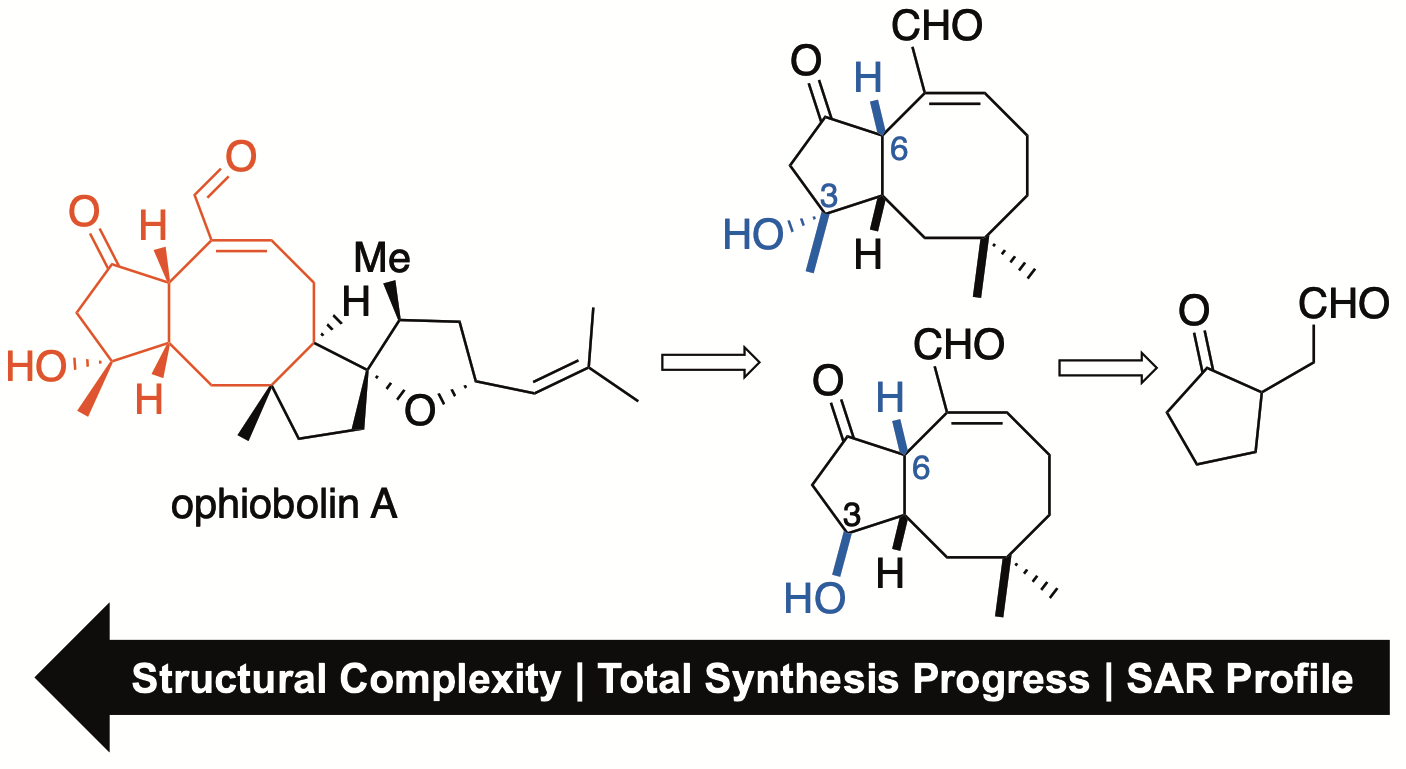
151. Pharmacophore-Directed Retrosynthesis Applied to Ophiobolin A: Simplified Bicyclic Derivatives Displaying Anticancer Activity Tao, Y.; Reisenauer, K.N.; Masi, M.; Evidente, A.; Taube, J.H.; Romo, D. Org. Lett. 2020, 22, 8307-8312.
150. Investigation of the mechanism of action of a potent pateamine A analog, des-methyl, des-amino pateamine A (DMDAPatA) Kommarahy, S. S.; Aulicino, J.; Gobbooru, S.; Li, J.; Zhu, M.; Romo, D.; Low, W. K. Biochem. Cell Biol. 2020, 98, 502-510.
149. Translation initiation factors GleIF4E2 and GleIF4A can interact directly with the components of the pre-initiation complex to facilitate translation initiation in Giardia lamblia Adedojaa, A.N.; McMahan, T.; Neal, J.P.; Dhakal, S.H.; Jois, S.; Romo, D.; Hull, K.; Garlapati, S. Mol. Biochem. Parasitol. 2020, 236, 111258.
148. Antiproliferative Activity of Naphthoquinones and Indane Carboxylic Acids from Lapachol Against a Panel of Human Cancer Cell Lines Eyong, K. O; Ketsemen, H. L.; Zhao, Z.; Du, L.; Ingels, A.; Mathieu, V.; Kornienko, A.; Hull, K. G.; Folefoc, G. N.; Baskaran, S.; Romo, D. Med. Chem. Res. 2020, 29, 1058–1066.

147. Generation and Reactivity of 2-Amido-1,3-diaminoallyl Cations: Cyclic Guanidine Annulations via Net (3 + 2) and (4 + 3) Cycloadditions. Kovvuri, V. R. R.; Xue, H.; Romo, D. Org. Lett. 2020, 22, 1407–1413.
2019
146. Molecular mechanism for attractant signaling to DHMA by E. coli Tsr Orr, A. A.; Yang, J.; Sule, N.; Chawla, R.; Hull, K. G.; Zhu, M.; Romo, D.; Lele, P. P.; Jayaraman, A.; Manson, M. D.; Tamamis, P. Biophys. J. 2019, 118, 492-504.
145. Novel Pateamine Analogs to Target the Translation Initiation Factor eIF4A in Chronic Lymphocytic Leukemia. Chen, R.; Zhu, M.; Chaudhari, R. R.; Robles, O.; Chen, Y.; Skillern, W.; Qin, Q.; Wierda, W.; Zhang, S.; Hull, K. G.; Romo, D.; Plunkett, W. Cancer Res. 2019, 79, 1854.
144. Pharmacophore-Directed Retrosynthesis Applied to Rameswaralide: Synthesis and Bioactivity of Sinularia Natural Product Tricyclic Cores. Truax, N.J.; Ayinde, S.; Van, K.; Liu, J.O.; Romo, D. Org. Lett. 2019, 21, 7394-7399.
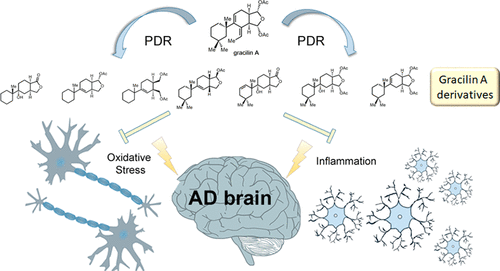
143. Gracilin A Derivatives Target Early Events in Alzheimer’s Disease: in Vitro Effects on Neuroinflammation and Oxidative Stress. Alvarino, R; Alonso, E.; Abbasov, M.E.; Chaheine, C.M.; Conner, M.L.; Romo, D.; Alfonso, A.; Botana, L.M. ACS Chem. Neurosci. 2019, 10, 4102-4111.
142. Natural Product Derivatization with β-Lactones, β-Lactams and Epoxides Toward 'Infinite' Binders. Jouanneau, M; Vellalath, S.; Kang, G.; Romo, D. Tetrahedron 2019, 75, 3348-3354.
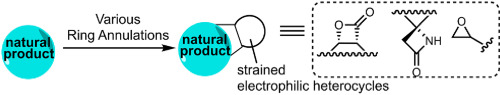
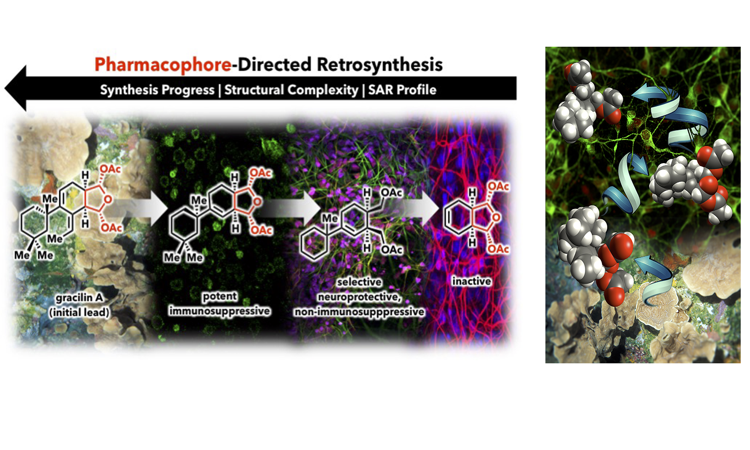
141. Simplified Immunosuppressive and Neuroprotective Agents Based on Gracilin A. Abbasov, M.E.; Alvarino, R.; Chaheine, C.M.; Alonso, E.; Sanchez, J.A.; Conner, M.L.; Alfonso, A.; Jaspars, M.; Botana, L.M.; Romo, D. Nat. Chem. 2019, 11, 342-350. (Click here for a full-text, view only version)
For further descriptions of this work, see: Sulikowski and Hudlicky: Nature News and Views Article (Click here for a full-text, view only version) and a Nature Research Chemistry Community 'Behind the Paper' Blog Post.
140. Creating Novel Translation Inhibitors to Target Pro-Survival Proteins in Chronic Lymphocytic Leukemia. Chen, R.; Zhu, M.; Chaudhari, R.R.; Robles, O.; Chen, Y.; Skillern, W.; Qin, Q.; Wierda, W.G.; Zhang, S.; Hull, K.G.; Romo, D.; Plunkett, W. Leukemia 2019, 33, 1663-1674.
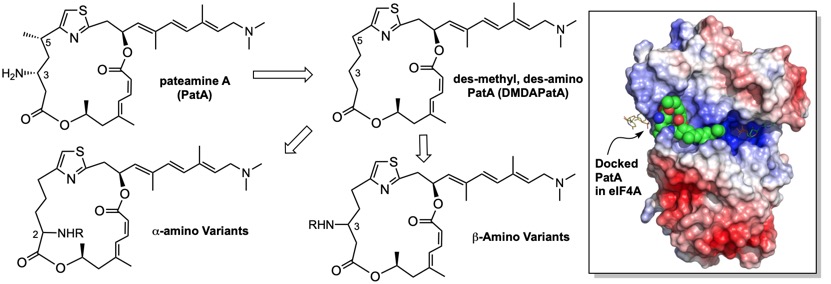
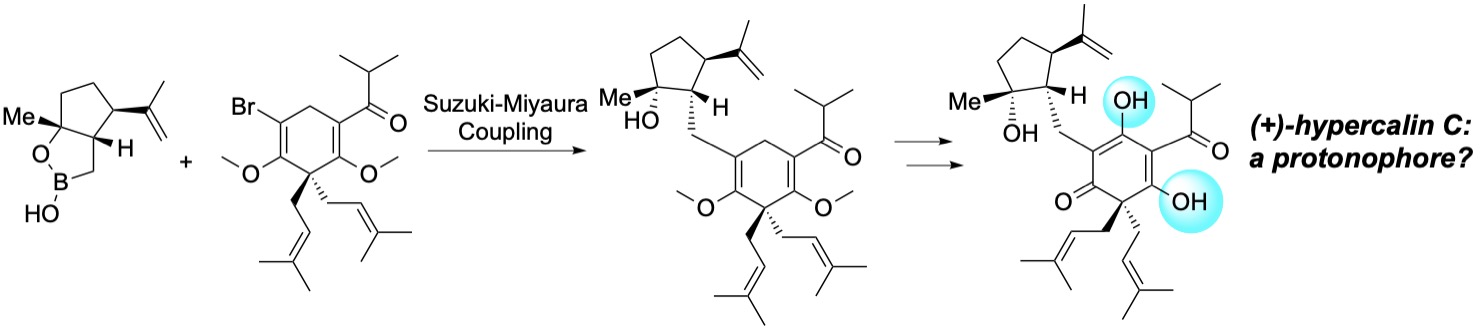
139. Total Synthesis and Anticancer Activity of (+)‐Hypercalin C and Congeners. Tao, Y.; Reisenauer, K.; Taube, J. H.; Romo, D. Angew. Chem. Int. Ed. 2019, 58, 2734-2738. [Featured in Synfacts]
138. A Novel Actin Binding Drug with In Vivo Efficacy. Ravichandran, A.; Geng, M.; Hull, K. G.; Li, J.; Romo, D. Lu, S-E.; Albee, A.; Nutter, C.; Gordon, D. M.; Ghannoum, M.A.; Lockless, S.W.; Smith, L. Antimicrob. Agents Chemother. 2019, 63, 1-13.
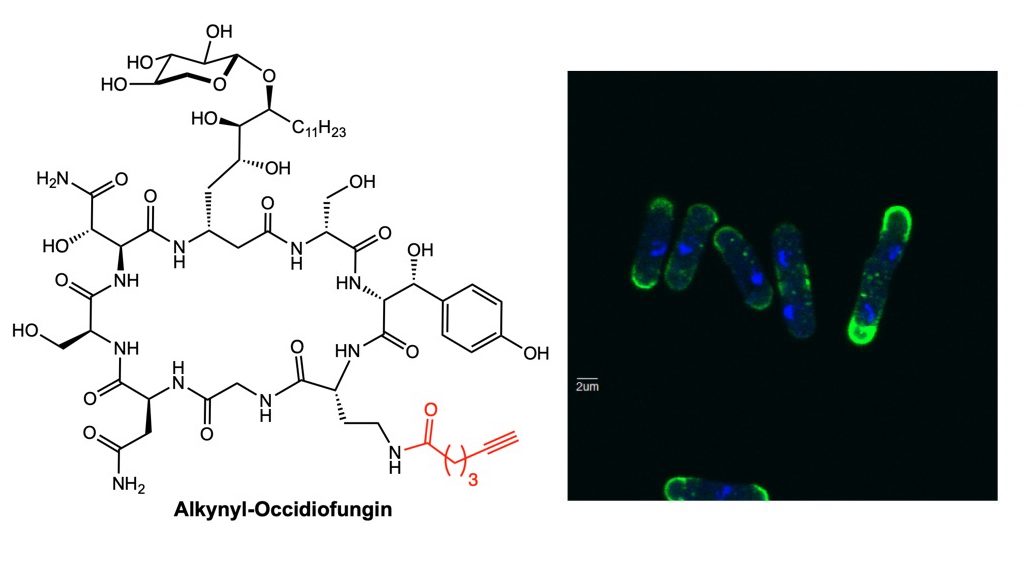
2018
137. (−)-Homosalinosporamide A and Its Mode of Proteasome Inhibition: An X-ray Crystallographic Study. Groll, M.; Nguyen, H.; Vellalath, S.; Romo, D. Mar. Drugs 2018, 16, 240.
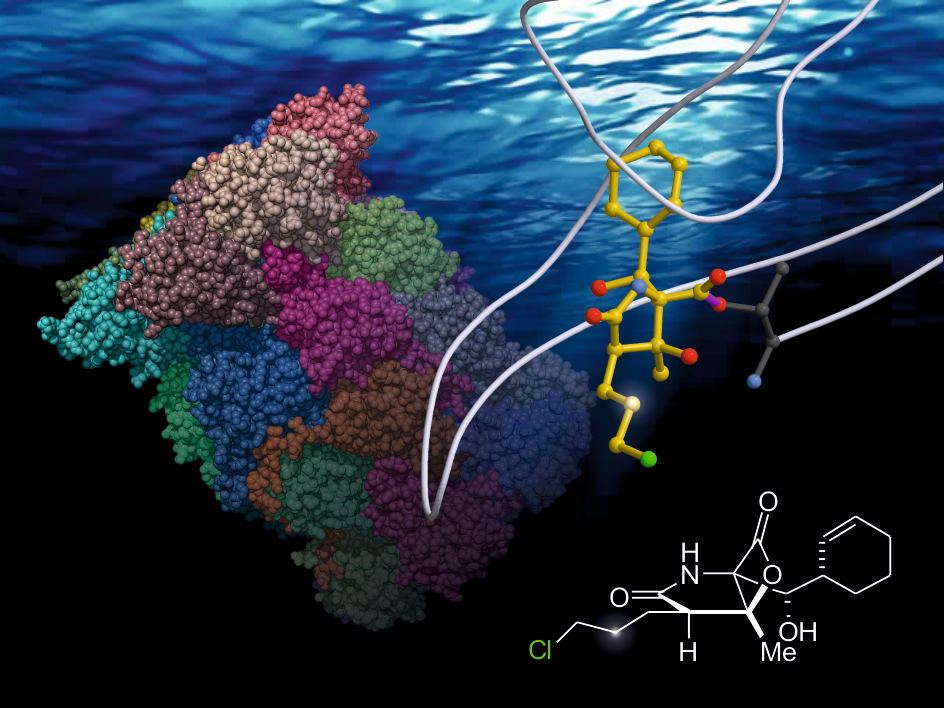
136. Enantioselective Synthesis of Medium-Sized Lactams via Chiral α,ß-Unsaturated Acylammonium Salts. Kang, G.; Yamagami, M.; Vellalath, S.; Romo, D. Angew. Chem. Int. Ed. 2018, 57, 6527–6531.

135. Isolation, Derivative Synthesis, and Structure-Activity Relationships of Anti-Parasitic Bromopyrrole Alkaloids from the Marine Sponge Tedania brasiliensis. Parra, L.; Bertonha, A.; Severo, I.; Aguiar, A.; de Souza, G.; Oliva, G.; Guido, R. V. C.; Grazzia, N.; Costa, T.; Miguel, D.; Gadelha, F.; Gilberto Ferreira, A.; Hajdu, E.; Romo, D.; Berlinck, R. J. Nat. Prod. 2018, 81, 188-202.

134. Multicomponent, Enantioselective Michael-Michael-Aldol-β-Lactonizations Delivering Complex β-Lactones. Van, K. N.; Romo, D. J. Org. Chem. 2018, 83, 632-643.
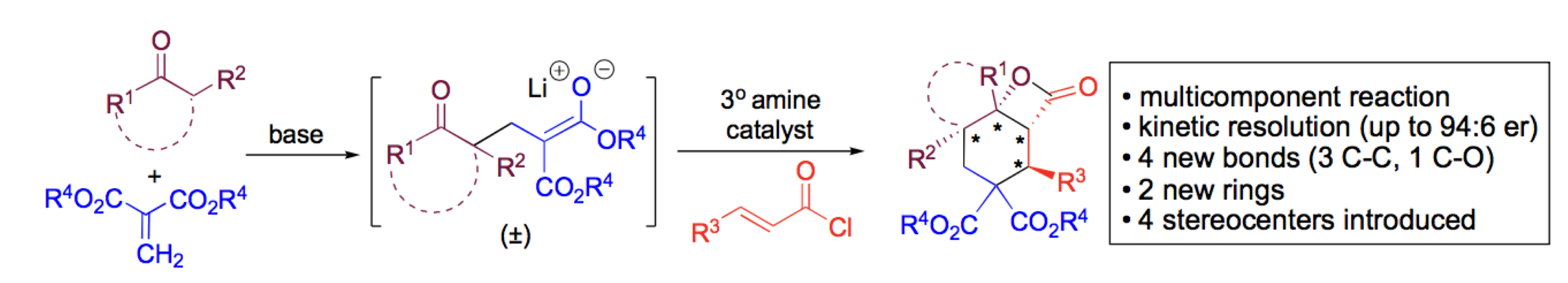
2017
133. Quantitative Chemoproteomic Profiling Reveals Multiple Target Interactions of Spongiolactone Derivatives in Leukemia Cells . Wright, M. H.; Tao, Y.; Drechsel, J.; Krysiak, J.; Chamni, S.; Weigert Muñoz, A.; Harvey, N.; Romo, D.; Sieber, S. A. Chem. Comm. 2017, 53, 12818-12821.
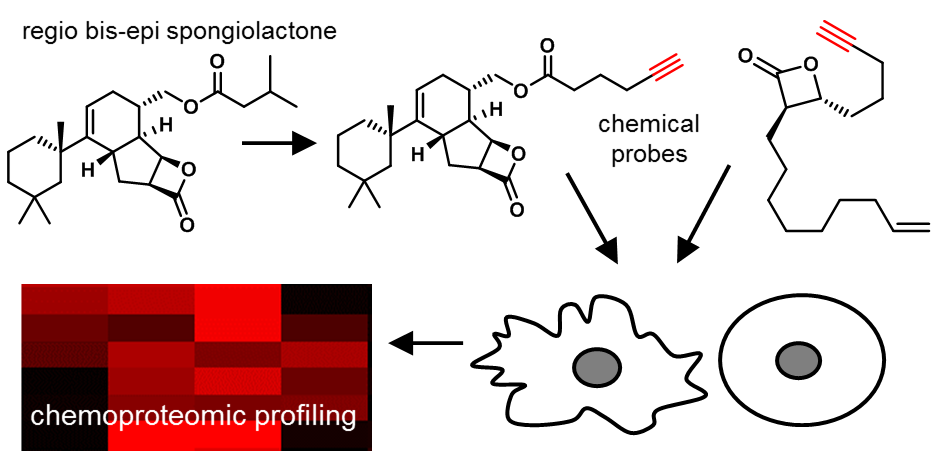
132. Silencing Oncogene Translation Using Pateamine A Anaolgues As a Novel Therapeutic Strategy for c-Myc Driven Lymphoma . Kim, Y.R.; Pal, I.; Lipstein, M. R.; Zhu, M.; Hull, K.G.; Romo, D.; O’Connor, O.A.; Deng, C. Blood 2017, 130, 4111.
131. Diastereo- and Enantioselective Synthesis of Bi- and Tricyclic N-Heterocycle-Fused β-Lactones. Kong, W.; Romo, D. J. Org. Chem. 2017, 82, 13161-13170. [Highlighted in Organic Chemistry Highlights].
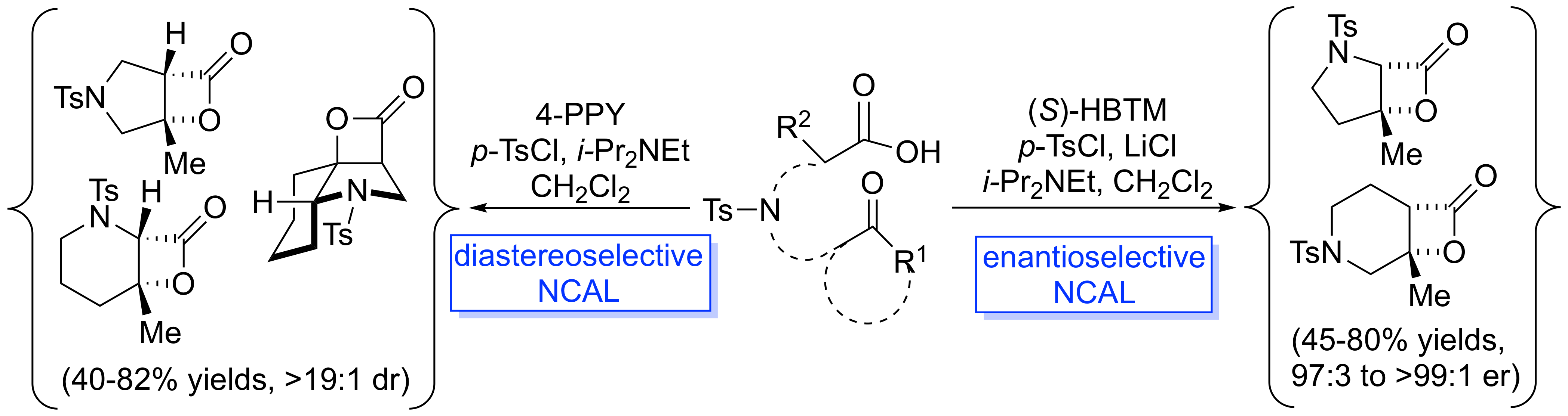
130. Bioactive Seco-Lanostane-Type Triterpenoids from the Roots of Leplaea mayombensis. Sidjui, L. S.; Eyong, K. O.; Hull, K. G.; Folefoc, G. N.; Leddet, V. M.; Herbette, G.; Ollivier, E.; Taube, J.; Klausmeyer, K.; Romo, D. J. Nat. Prod. 2017, 80, 2644–2651.
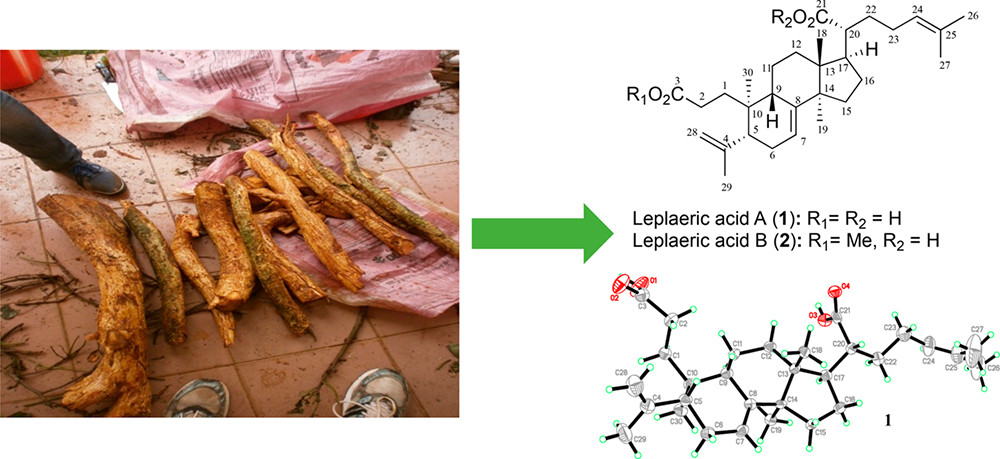
129. Triterpenoids from the stem bark of Vitellaria paradoxa (Sapotaceae) and derived esters exhibit cytotoxicity against a breast cancer cell line. Eyong, K. O; Baïrys, G.; Eno, A. A.; Taube, J.; Hull, K. G.; Folefoc, G. N.; Foyet, H. S.; Romo, D. Med. Chem. Res. 2017, 27, 268-277.
128. Inhibition of Eukaryotic Translation by the Antitumor Natural Product Agelastatin A . McClary, B.; Boris, Z.; Meyer, M.; Jouanneau, M.; Pellegrino, S.; Yusupova, G.; Schuller, A.; Reyes, J. C.; P., Lu, J.; Luo, C.; Dang, Y.; Romo, D.; Yusupov, M.; Green, R.; Liu, J. O. Cell Chem. Biol. 2017, 24, 605-613.
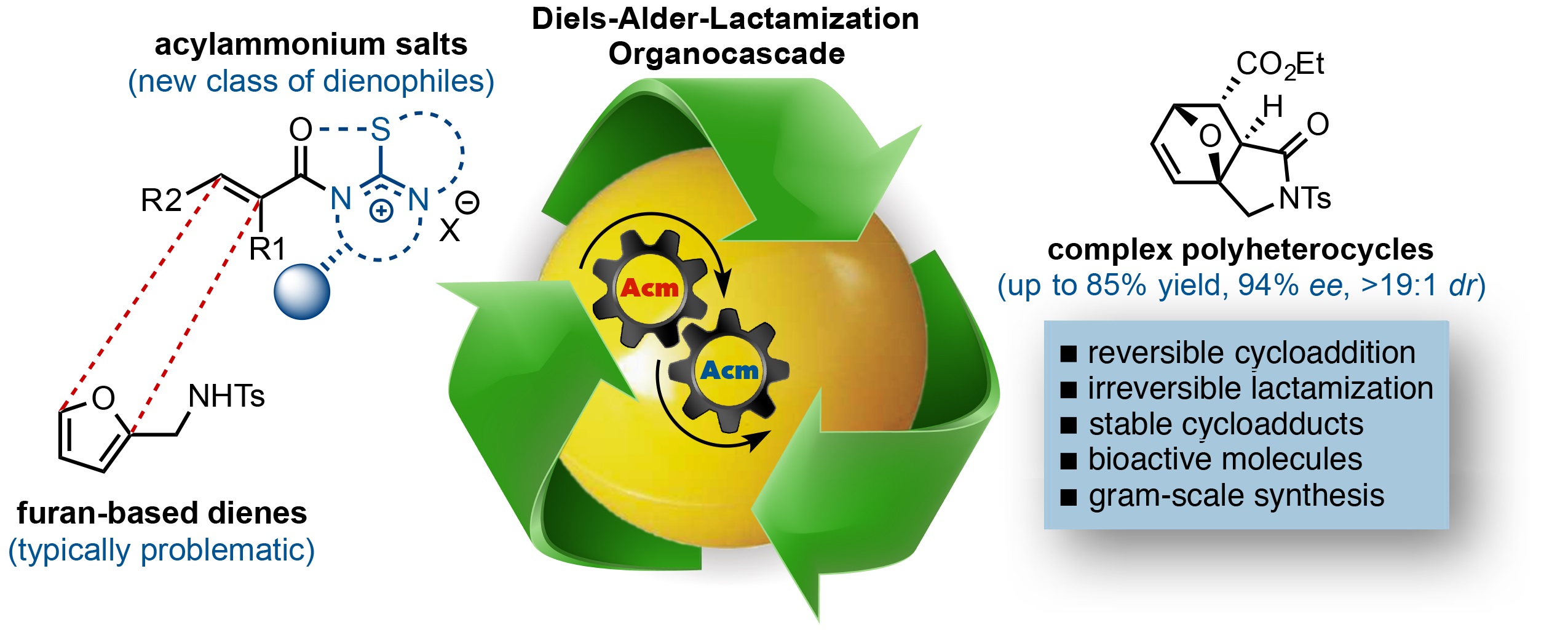
127. Enantioselective Diels-Alder-Lactamization Organocascades Employing a Furan-Based Diene Abbasov, M. E.; Hudson, B. M.; Kong, W.; Tantillo, D. J.; Romo. D. Org. Biomol. Chem. 2017, 33, 592-594.
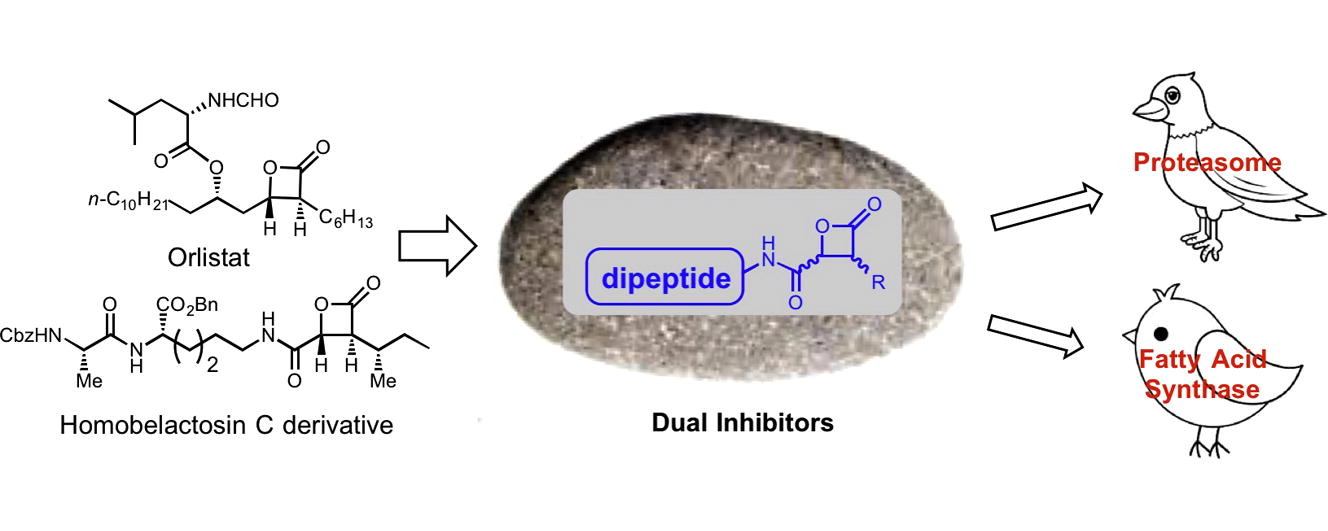
126. A Strategy for Dual Inhibition of the Proteasome and Fatty Acid Synthase with Belactosin C-Orlistat Hybrids . Zhu, M.; Harshbarger, W.; Robles, O.; Krysiak, J.; Hull, K.; Cho, C. S; Richardson, R.; Yang, Y.; Garcia, A.; Spiegelman, L.; Ramirez, B.; Wilson, C.; Yau, J. A.; Moore, J.; Walker, C.; Sacchettini, C; Liu, W.; Sieber, S.; Smith, J; Romo, D. Bioorg. Med. Chem. Lett. 2017. 25, 2901-2919.
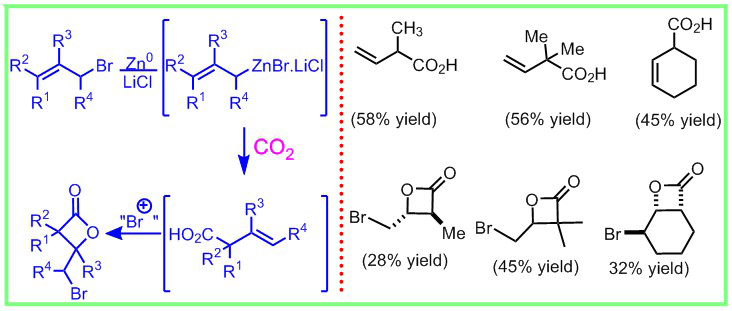
125. Telescoped Synthesis of γ-Bromo-β-Lactones from Allylic Bromides Employing Carbon Dioxide Vellalath, S.; Romo, D., Isr. J. Chem. 2017, 57, 1-6. [Highlighted in Wiley-VCH Hot Topics: CO2].
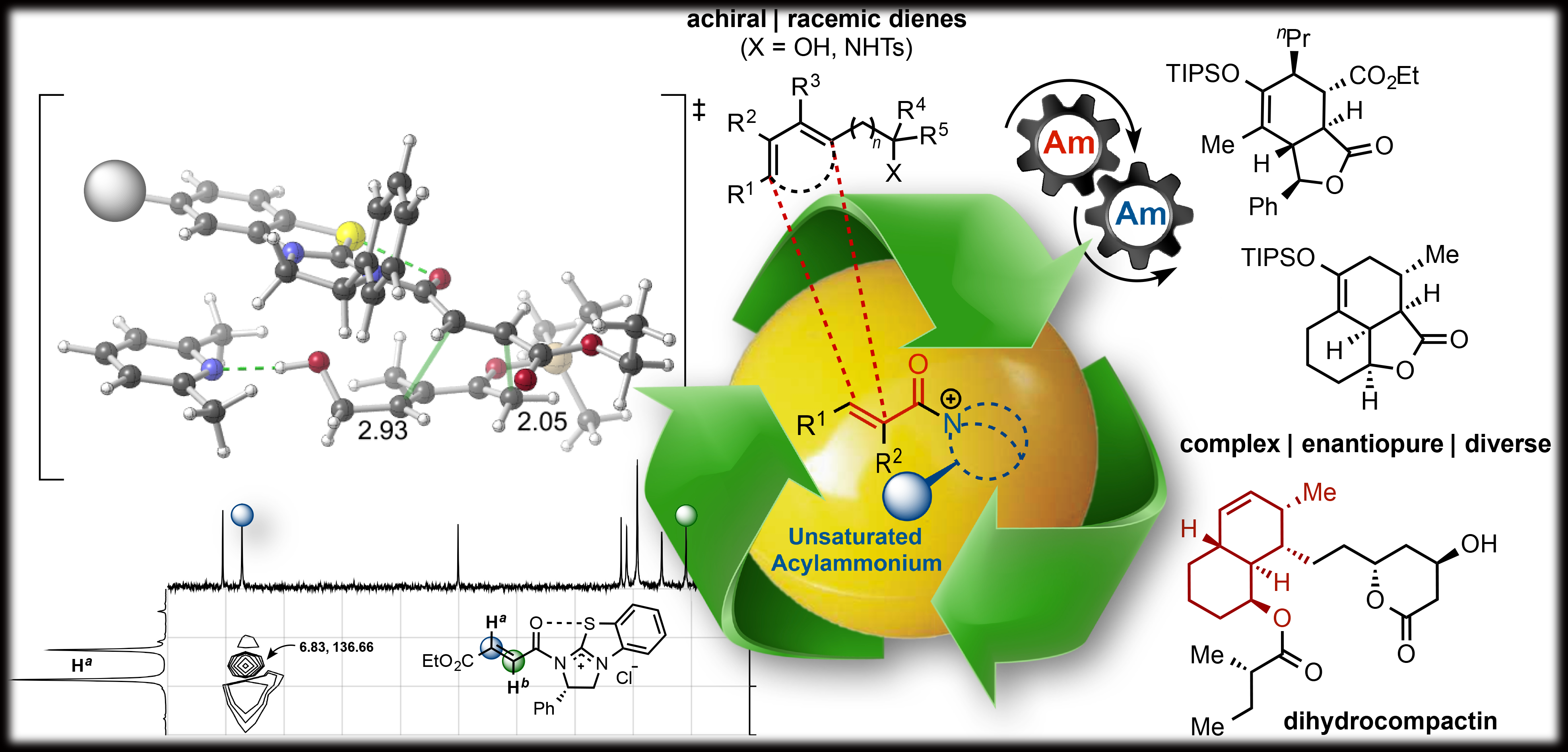
124. Stereodivergent, Diels‐Alder‐Initiated Organocascades Employing α,β-Unsaturated Acylammonium Salts: Scope, Mechanism, and Applications Abbasov, M. E.; Hudson, B. M.; Tantillo, D. J.; Romo D., Chem. Sci. 2017, 8, 1511-1524.
2016
123. Catalytic Generation of Ammonium Enolates and Related Tertiary Amine-Derived Intermediates: Applications, Mechanism, and Stereochemical Models (n→π*) Van, K. N.; Morrill, L. C.; Smith, A. D.; Romo, D.,In Lewis Base Catalysis in Organic Synthesis; Vedejs, E.; Denmark, S. E., Eds.; Wiley-VCH: 2016. (Book chapter).
122. Editorial: Strategies for cellular target identification of natural products J. O. Liu and D. Romo, Nat. Prod. Rep. 2016, 33, 592-594.

121. The Emerging Utility of α,β-Unsaturated Acylammonium Salts for Asymmetric Organocatalysis Vellalath, S.; Romo, D. Angew. Chem. Int. Ed. 2016, 55, 13934-13943.
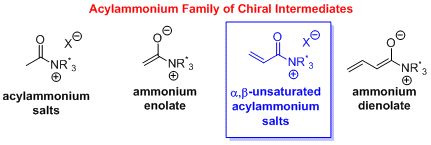
120. Chemical Mechanism of the Phosphotriesterase from Sphingobiumsp. Strain TCM1, an Enzyme Capable of Hydrolyzing Organophosphate Flame Retardants Bigley, A. N.; Xiang, D. F.; Ren, Z.; Xue, H.; G. Hull, K. G.; Romo, D.; Raushel, F. M. J. Am. Chem. Soc. 2016, 138, 2921-2924.
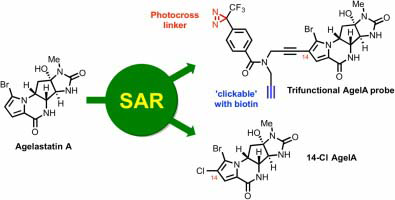
119. Derivatization of Agelastatin A Leading to Bioactive Analogs and a Trifunctional Probe . Jouanneau, M.; McClary, B.; Reyes, J. C. P.; Chen, R.; Chen, Y.; Plunkett, W.; Cheng, X.; Milinichik, A. Z.; Albone, E. F.; Liu, J. O.; Romo, D. Bioorg. Med. Chem. Lett. 2016, 26, 2092-2097.
2015
118. Piperlongumine Blocks JAK2-STAT3 to Inhibit Collagen-Induced Platelet Reactivity Independent of Reactive Oxygen Species . Yuan, H.; Houck, K. L., Tian, Y.; Bharadwaj, U.; Hull, K.; Zhou, M.; Wu, X.; Tweardy, D. J.; Romo, D.; Fu, X.; Zhang Y.; Zhang, J.; Dong, J.-F. PLOS one 2015, 10, e0143964.
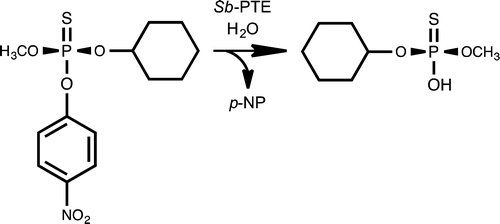
117. Interrogation of the Substrate Profile and Catalytic Properties of the Phosphotriesterase from Sphingobium sp. Strain TCM1: An Enzyme Capable of Hydrolyzing Organophosphate Flame Retardants and Plasticizers. Xiang D. F.; Bigley, A. N.; Zhongjie Ren, Z.; Xue, H.; Hull K. G.; Romo, D.; Frank M. Raushel, F. M. Biochemistry 2015, 54, 7539-7549.
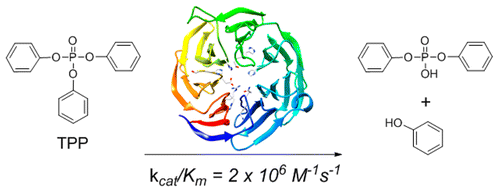
116. Utility and NMR Studies of α,β-Unsaturated Acylammonium Salts: Synthesis of Polycyclic Dihydropyranones and a Dihydropyridone Vellalath, S.; Van, K. N.; Romo, D. Tetrahedron Lett. 2015 23, 3647–3652. (Commemorative Issue for the late Prof. Harry Wasserman).
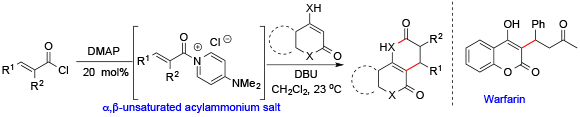
115. Synthesis of (±)-Spongiolactone Enabling Discovery of a More Potent Derivative Harvey, N. L.; Krysiak, J.; Chamni, S.; Cho, S. W.; Sieber, S. A.; Romo, D. Chem. Eur. J. 2015, 21, 1425–1428.
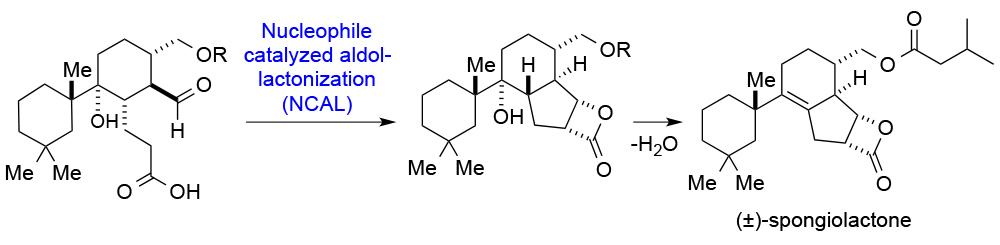
114. Covalent Modification of a Cysteine in XPB Through Single Epoxide Cleavage of the Transcription Inhibitor Triptolide He, Q.-L.; Titov, D. V.; Li, J.; Tan, M.; Ye, Z.; Zhao, Y.; Romo, D.; Liu, J. O. Angew. Chem. Int. Ed. 2015, 54, 1859–1863.
2014
113. 2-Bromo-N-n-propyl-pyridinium-1,1,1-trifluoromethanesulfonate Jouanneau, M.; Romo, D. Encyl. Reag. Org. Syn. 2014, 1–2.
112. Role of an Active Site Loop in the Promiscuous Activities of Amycolatopsis sp. T-1-60 NSAR/OSBS McMillan, A. W.; Lopez, M. S.; Zhu, M.; Morse, B. C.; Yeo, I.-C.; Amos, J.; Hull, K.; Romo, D.; Glasner, M. E. Biochemistry 2014, 53, 4434–4444.
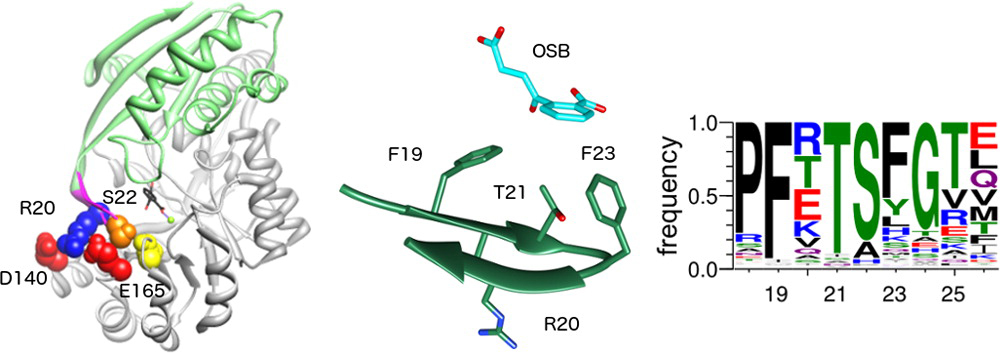
111. Promiscuity of Exiguobacterium sp. AT1b o-Succinylbenzoate Synthase Illustrates Evolutionary Transitions in the OSBS Family Brizendine, A. M.; Odokonyero, D.; McMillan, A. W.; Zhu, M.; Hull, K.; Romo, D.; Glasner, M. E. Biochem. Biophys. Res. Commun. 2014, 450, 679–684.
110. The Ever-Expanding Role of Asymmetric Covalent Organocatalysis in Scalable, Natural Product Synthesis Abbasov, M. E.; Romo D. Nat. Prod. Rep. 2014, 31, 1318–1327.
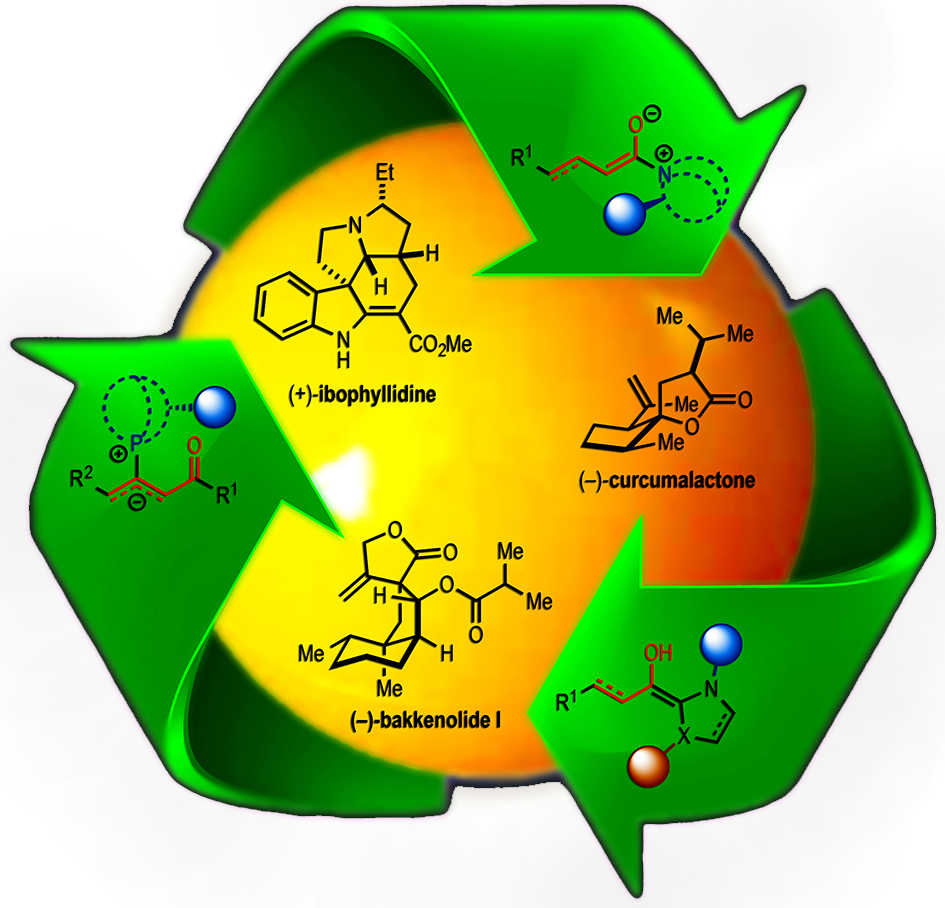
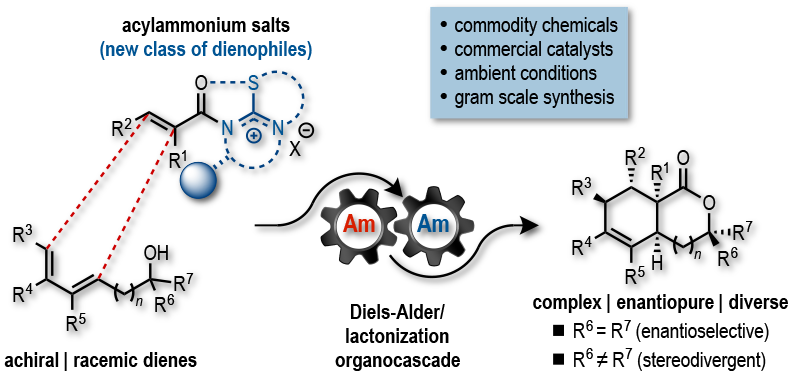
109. Acylammonium Salts as Dienophiles in Diels-Alder/Lactonization Organocascades Abbasov, M. E.; Hudson, B. M.; Tantillo, D. J.; Romo D. J. Am. Chem. Soc. 2014, 136, 4492–4495. [Featured in JACS Spotlights, Synfacts, and Advances in Engineering]
108. Chemo- and Site-Selective Derivatizations of Natural Products Enabling Biological Studies Robles, O.; Romo D. Nat. Prod. Rep. 2014, 31, 318-334.
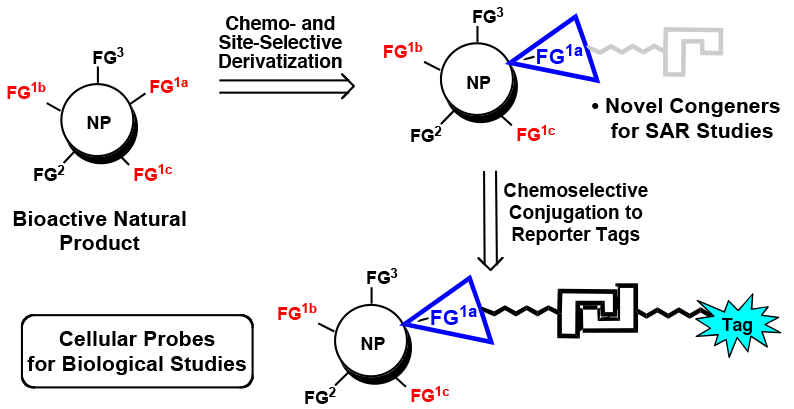
107. Second-Generation Derivatives of the Eukaryotic Translation Initiation Inhibitor Pateamine A Targeting eIF4A as Potential Anticancer Agents Low, W.-K.; Li, J.; Zhu, M.; Kommaraju, S. S.; Shah-Mittal, J.; Hull, K.; Liu, J. O.; Romo D. Bioorg. Med. Chem. Lett. 2014, 22, 116-125.
2013
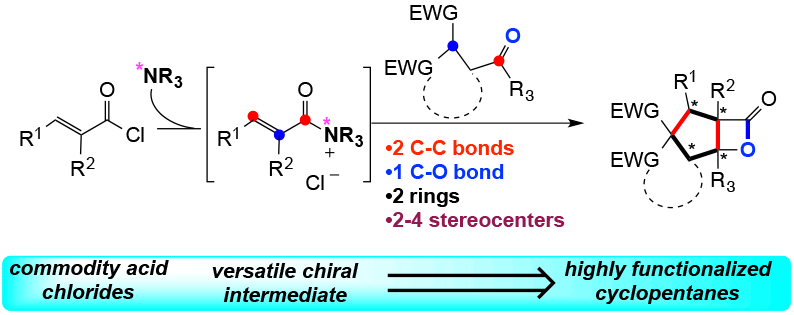
106. Rapid Assembly of Complex Cyclopentanes Employing Chiral, α,β-Unsaturated Acylammonium Intermediates Liu, G.; Shirley, M. E.; Van, K. N.; McFarlin, R. L.; Romo, D. Nat. Chem., 2013, 5, 1049-1057. [Highlighted in C&EN, BioNewsTexas, and Synform]
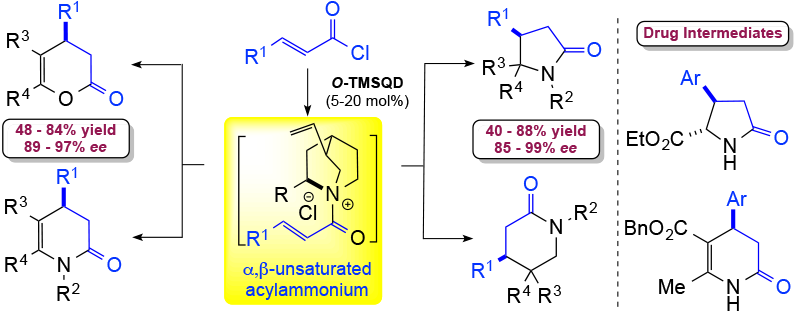
105. Direct Catalytic Asymmetric Synthesis of N-Heterocycles from Commodity Acid Chlorides Employing α,β-Unsaturated Acylammoniums Vellalath, S.; Van, K. N.; Romo, D. Angew. Chem. Int. Ed., 2013, 52, 13688–13693. [Highlighted in Synfacts (heterocycles) and Synfacts (organocatalysis)]
104. Interrogating Translational Efficiency and Lineage-Specific Transcriptomes Using Ribosome Affinity Purification Zhou, P.; Zhang, Y.; Ma, Q.; Gu, F. Day, D. S.; He, A.; Zhou, B., Li, J., Stevens, S.; Romo, D.; Pu, W. T. Proc. Nat. Acad. Sci., 2013, 110, 15395-15400.
103. Concise Synthesis of the Isothiourea Organocatalysts Homobenzotetramisole and Derivatives Ranieri, B.; Robles, O.; Romo, D. J. Org. Chem., 2013, 78, 6291-6296.
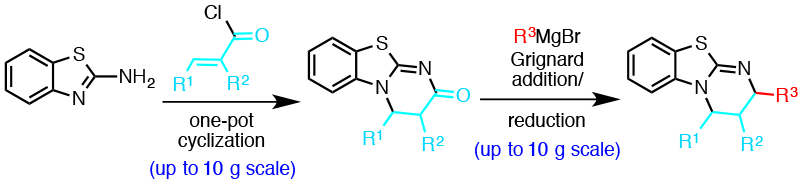
102. Simultaneous Structure–Activity Studies and Arming of Natural Products by C–H Amination Reveal Cellular Targets of Eupalmerin Acetate Li, J.; Cisar, J.S.; Zhou, C.-Y.; Vera, B.; Williams, H.; Rodrı´guez, A.B.; Cravatt, B.F.; Romo, D. Nat. Chem., 2013, 5, 510-517.
2012
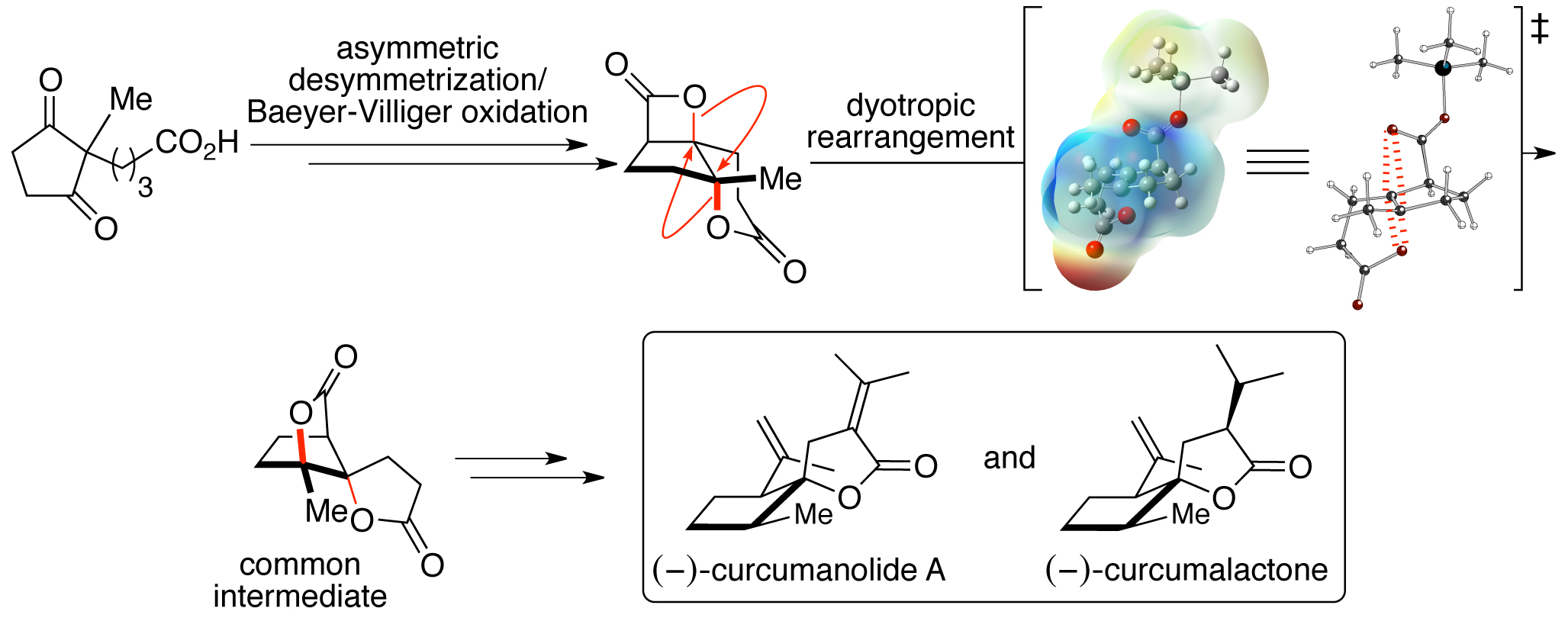
101. Dyotropic Rearrangements of Fused Tricyclic β-Lactones: Application to the Synthesis of (−)-Curcumanolide A and (−)-Curcumalactone Leverett C.A.; Purohit V.C.; Johnson A.G.; Davis R.L.; Tantillo D.J.; Romo D. J. Am. Chem. Soc., 2012, 134, 13348-13356.
100. Bioinspired Total Synthesis of Agelastatin A Reyes, J.C.P.; Romo, D. Angew. Chem. Int. Ed., 2012, 51, 6870-6873.

99. De Novo Synthesis of Benzosceptrin C and Nagelamide H from 7-15N-Oroidin: Implications for Pyrrole–Aminoimidazole Alkaloid Biosynthesis Stout E.P.; Morinaka, B.I.; Wang, Y.-G.; Romo, D.; Molinski, T.F. J. Nat. Prod., 2012, 75, 527-530.
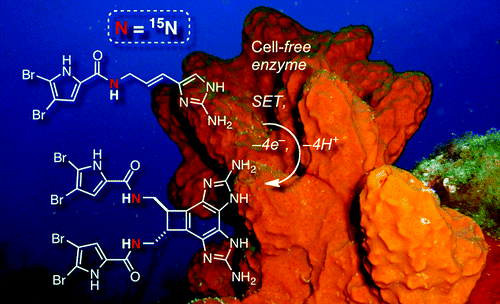
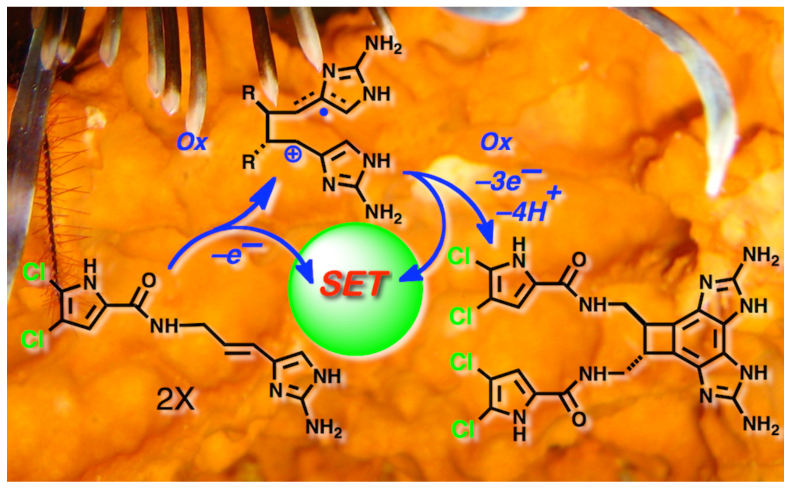
98. Pyrrole-Aminoimidazole Alkaloid Metabiosynthesis with Marine Sponges Agelas Conifera and Stylissa Caribica Stout, E.P.; Wang, Y.-G.; Romo, D.; Molinski, T.F. Angew. Chem. Int. Ed., 2012, 51, 4877-4881.
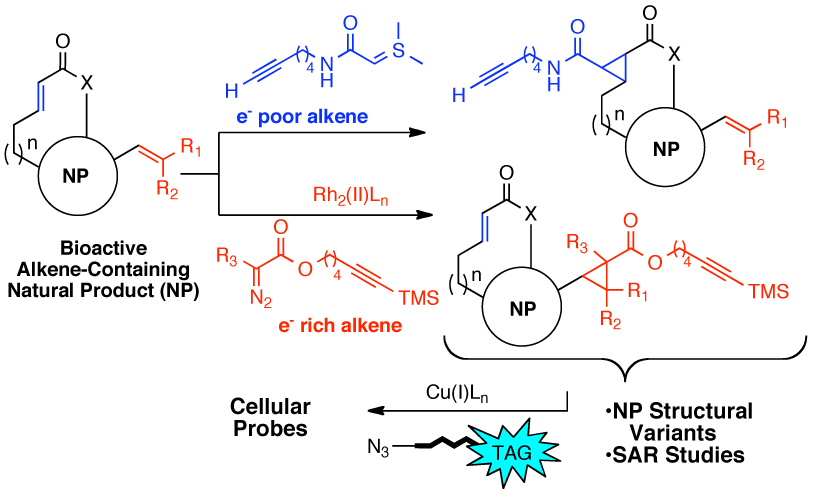
97. Cyclopropanations of Olefin-Containing Natural Products for Simultaneous Arming and Structure Activity Studies Robles, O.; Sergio O. Serna-Saldívar, S.O.; Gutiérrez-Uribe, J.A.; Romo, D. Org. Lett., 2012, 14, 1394-1397.
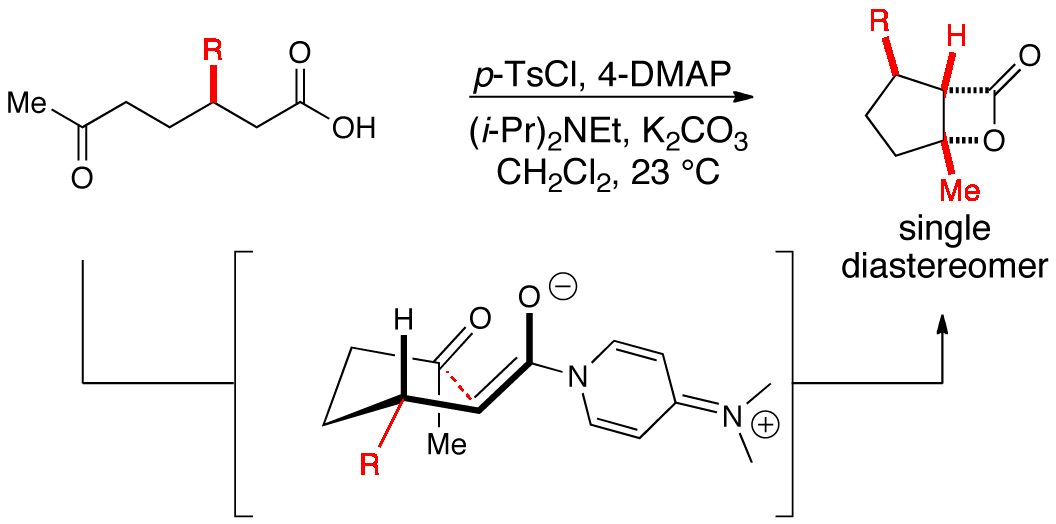
96. A Diastereoselective, Nucleophile-Promoted Aldol-Lactonization of Ketoacids Leading to Bicyclic-β-Lactones Liu, G.; Shirley, M.E.; Romo, D. J. Org. Chem., 2012, 77, 2496-2500.
2011
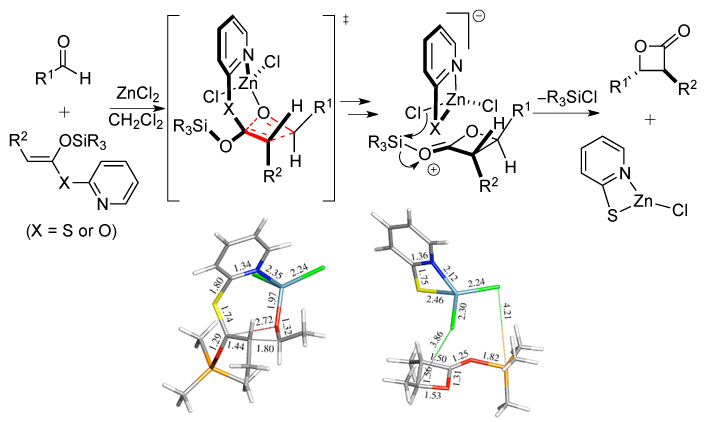
95. Mechanistic Investigations of the ZnCl2-Mediated Tandem Mukaiyama Aldol-Lactonization (TMAL): Evidence for Asynchronous, Concerted Transition States and Discovery of 2-Oxopyridyl Ketene Acetal Variants Zhao, C.; Mitchell, T. A.; Vallakati, R.; Pérez, L. M. Romo, D. J. Am. Chem. Soc., 2011, 134, 3084-3094.
94. Total Synthesis of the Spirocyclic Imine Marine Toxin (-)-Gymnodimine and an Unnatural C4-Epimer Kong, K.; Moussa, Z.; Lee, C.; Romo, D. J. Am. Chem. Soc., 2011, 133, 19844-19856. [Highlighted by Chemistry World in Totally Synthetic (-)-Gymnodimine]
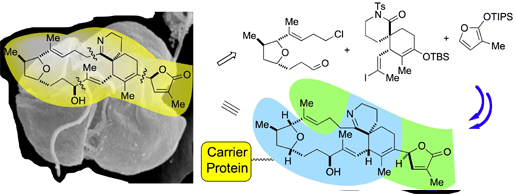
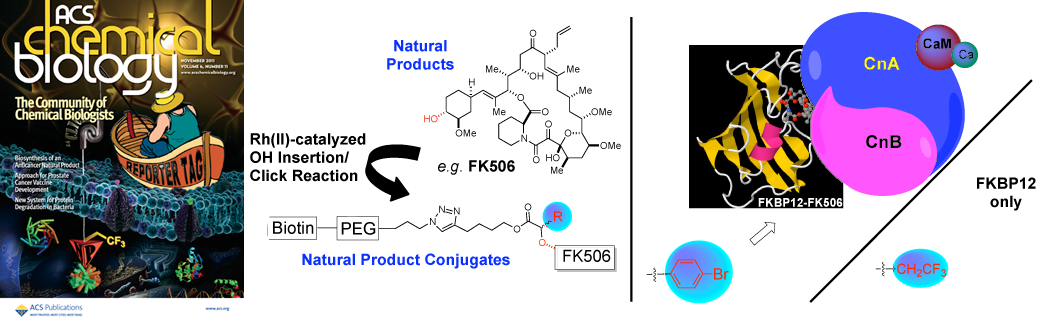
93. Diazo Reagents with Small Steric Footprints for Simultaneous Arming/SAR Studies of Alcohol-Containing Natural Products via OH Insertion Chamni, S.; He, Q.L.; Dang, Y.; Bhat, S.; Liu, J.O.; Romo, D. ACS Chem. Biol., 2011, 6, 1175-1181. [Featured on Issue 11, 2011 Front Cover]
92. Switching Between Concerted and Stepwise Mechanisms for Dyotropic Rearrangements of β-Lactones Leading to Spirocyclic, Bridged γ-Butyrolactones Davis, R.; Leverett, C.; Romo, D.; Tantillo, D. J. Org. Chem., 2011, 76, 7167-7174.

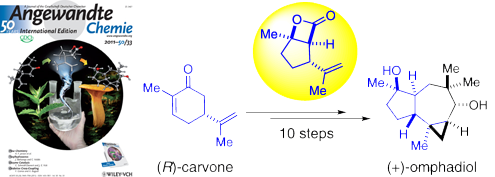
91. Total Synthesis of (+)-Omphadiol Liu, G.; Romo, D. Angew. Chem. Int. Ed., 2011,123, 7679-7682. [Featured on Issue 33, 2011 Front Cover and Highlighted in Synfacts 2011, 10, 1043]
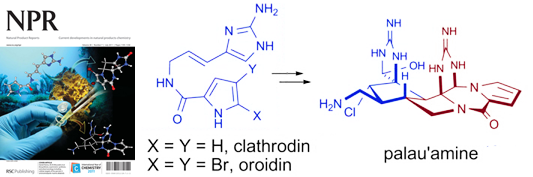
90. Biosynthesis, Asymmetric Synthesis, and Pharmacology Including Cellular Targets of the Pyrrole-2-Aminoimidazole Marine Alkaloid Al-Mourabit, A.; Zancanella, M.; Tilvi, S.; Romo, D. Nat. Prod. Rep., 2011, 28, 1229-1260. [Featured on Issue 7, 2011 Front Cover]
89. Organocatalytic Enantioselective Synthesis of Bicyclic β-Lactones Via Nucleophile-Catalyzed Aldol-Lactonization (NCAL) Nguyen, H.; Oh, S.; Henry-Riyad, H.; Sepulveda, D.; Romo, D. Org. Syn., 2011, 88, 121-137.
88. Marinobufagenin Levels in Preeclamptic Patients: A Preliminary Report Horvat, D.; Harrison, R.; Uddin, M.N.; Jones, R.; Abi Ghanem, D.; Berghman, L.C.; Lai, X.Z.; Li, J.; Romo, D.; Puschett, J.B. Amer. J. Perinatology, 2011, 28, 509-514.
87. A Chemifluorescent Immunoassay for the Determination of Marinobufagenin in Body Fluids Abi-Ghanem, D.; Lai, X.Z.; Berghman, L.R.; Horvat, D.; Li, J.; Romo, D.; Uddin, M.N.; Kamano, Y .; Nogawa, T.; Xu, J.P.; Pettit, G.R.; Puschett, J.B. J. Immunoassay Immunochem., 2011, 32, 31-46.
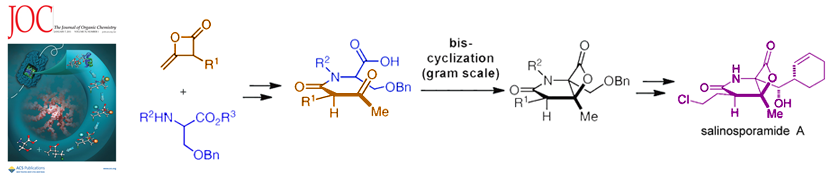
86. Bioinspired Total Synthesis and Human Proteasome Inhibitory Activity of (-)-Salinosporamide A, (-)-Homosalinosporamide A, and Derivatives Obtained via Organonucleophile Promoted Bis-Cyclizations Nguyen, H.; Ma, G.; Fremgen,T.; Gladysheva, T.; Romo, D. J. Org. Chem., 2011, 76, 2-12. [Featured on Issue 1, 2011 Front Cover]
2010
85. Enantioselective, Organocatalyzed Intramolecular Aldol-Lactonizations with Ketoacids Leading to Bicyclic and Tricyclic β-Lactones and Topology Morphing Transformations Leverett, C.A.; Purohit, V.C.; Romo, D. Angew. Chem. Int. Ed., 2010, 49, 9479-9483. [Highlighted in Synfacts 2011, 1, 99]
84. Cyclosporin A Treatment of Leishmania Donovani Reveals Stage-Specific Functions of Cyclophilins in Parasite Proliferation and Viability Yau, W.L.; Blisnick, T.; Taly, J.F.; Helmer-Citterich, M.; Schiene-Fischer, C.; Leclercq, O.; Li, J.; Schmidt-Arras, D.; Morales, M.A.; Notredame, C.; Romo, D.; Bastin, P.; Spath, G.F. Plos Negl. Trop. Dis., 2010, 4, e729.
83. Double Diastereoselective, Nucleophile-Catalyzed Aldol Lactonizations (NCAL) Leading to β-Lactone Fused Carbocycles and Extensions to β-Lactone Fused Tetrahydrofurans Morris, K. A.; Arendt, K.; Oh, S.; Romo, D. Org. Lett., 2010, 12, 3764-3767.
82. A1,3-Strain Enabled Retention of Chirality During Bis-Cyclization of β-Ketoamides: Total Synthesis of (–)-Salinosporamide A and (–)-Homosalinosporamide A Nguyen, H.; Ma, G.;Romo, D. Chem. Comm., 2010, 46, 4803-4805.
81. Mild Arming and Derivatization of Natural Products via an In(OTf)3-Catalyzed Arene Iodination Zhou, C-Y; Li, J.; Peddibhotla, S.; Romo, D. Org. Lett., 2010, 12, 2104-2107.
80. Synthesis of 7-15N-Oroidin and Evaluation of Utility for Biosynthetic Studies of Pyrrole-Imidazole Alkaloids by Microsale 1H-15N HSQC and FTMS Wang, Y.; Morinaka, B.I.; Reyes, J.C.P.; Wolff, J. H.; Romo, D.; Molinski, T. F. J. Nat. Prod., 2010, 73, 428-434.
2009
79. Enantioselective Total Synthesis of the Marine Toxin (-)-Gymnodimine Employing a Barbier-Type Macrocyclization Kong, K.; Lee, C.S.; Romo, D. Agnew. Chem. Int. Ed., 2009, 121, 7538-7541. [Highlighted in Synfacts 2010, 1, 15]
78. Inhibition of Nonsense-mediated mRNA Decay by the Natural Product Pateamine A Through Eukaryotic Initiation Factor 4AIII Dang, Y.; Low, W-K.; Xu, J.; Gehring, N. H.; Dietz, H.C.; Romo, D.; Liu, J.O. J. Biol. Chem., 2009, 35, 23613-23621.
77. Asymmetric Synthesis, Structure, and Reactivity of Unexpectedly Stable Spiroepoxy-β-Lactones Including Facile Conversion to Tetronic Acids: Application to (+)-Maculalactone A Duffy, R. J.; Morris, K.A.; Vallakati, R.; Zhang, W.; Romo, D. J. Org. Chem., 2009, 74, 4772-4781.
76. Synthesis of Unusually Strained Spiro Ring Systems and Their Exploits in Synthesis Duffy, R. J.; Morris, K. A.; Romo, D. Tetrahedron 2009, 65, 5879-5892.
75. Potent In Vitro and In Vivo Anticancer Activities of Des-methyl, Des-amino Pateamine A, a Synthetic Analogue of Marine Natural Product Pateamine A Kuznetsv, G.; Xu,Q.;Rudolph-Owen,L.; TenDyke, K.; Liu, J.; Towle, M.; Zhao, N.; Marsh, J.; Agoulnik, S.; Twine, N.; Parent, L.; Chen, Z.; Shie, J.; Jian, Y.; Zhang, H.; Du, H.; Boivin, R.; Wang, Y.; Romo, D.; Littlefield, B. Mol Cancer Ther., 2009, 8, 1250-1260.
74. Enantioselective Synthesis of Schulzeines B and C via a ß-Lactone-Derived Surrogate for Bis-homoserine Aldehyde Romo, D.; Liu, G. Org. Lett., 2009, 11, 1143-1146.
2008
73. Diastereoselective, Three-Component, Cascade Synthesis of Tetrahydrofurans and Tetrahydropyrans Employing the Tandem Mukaiyama Aldol-Lactonization (TMAL) Process Mitchell, T. A.; Zhao, C.; Romo, D. J. Org. Chem., 2008, 73, 9544-9551.
72. Recent Advances in Lewis Base-Catalyzed, Stereoselective Aldol β- and γ- Lactonizations Purohit, V. C.; Matla, A. S.; Romo, D. Heterocycles 2008, 76, 949-979.
71. Concise Synthesis of Spirocyclic, Bridged γ-Butrolactones via Stereospecific, Dyotropic Rearrangements of β-Lactones Involving 1,2-Acyl and δ-Lactone Migrations Purohit, V. C.; Matla, A. S.; Romo, D. J. Am. Chem. Soc., 2008, 130, 10478-10479.
70. Synthesis of Novel β-Lactone Inhibitors of Fatty Acid Synthase Richardson, R. D.; Ma, G.; Oyola, Y.; Zancanella, M. A.; Knowles, L. M.; Cieplak, P.; Romo, D.; Smith, J. W. J. Med. Chem., 2008, 51, 5285-5296.
69. Facile Sythesis of the Trans-Fused Azabicyclo[3.3.0]octane Core of Palau’amine and the Tricyclic Core of Axinellamine from a Common Intermediate Zancanella, M. A.; Romo, D. Org. Lett., 2008, 10, 3685-3688.
68. Highly Diastereoselective, Tandem, Three-Component Synthesis of Tetrahydrofurans from Ketoaldehydes via Silylated β-Lactone Intermediates: Application to a Colpsinol B Fragment Mitchell, T. A.; Zhao, C.; Romo, D. Angew. Chem. Int. Ed., 2008, 47, 5026-5029.
67. β-Lactam Congeners of Orlistat as Inhibitors of Fatty Acid Synthase Zhang, W.; Richardson, R. D.; Chamni, S.: Smith, J. W.; Romo, D. Bioorg. Med. Chem. Lett., 2008, 28, 2491-2494. [Highlighted in C&EN, June 23, 2008]
66. Enantioselective Synthesis of (+)-Monobromophakellin and (+)-Phakellin: A Concise Phakellin Annulation Strategy Applicable to Palau'amine Wang, S.; Romo, D. Angew. Chem. Int. Ed., 2008, 47, 1284-1286. [Highlighted in Synfacts 2008, 7, 678]
2007
65. Synthesis of a Cyclic Guanidine Hemiaminal Pertinent to the Axinellamines Tang, L.; Romo, D. Heterocycles 2007, 74, 999-1008.
64. Diastereoselective Synthesis of Tetrahydrofurans via Mead Reductive Cyclization of Keto-β-Lactones Derived from the Tandem Mukaiyama Aldol Lactonization (TMAL) Process Mitchell, T. A.; Romo, D. J. Org. Chem., 2007, 72, 9053-9059. [Highlighted in Organic Chemistry Portal: Organic Chemistry Highlights, April 14, 2008]
63. Transformation of Fused Bicyclic and Tricyclic β-Lactones to Fused γ-Lactones and 3(2H)-Furanones via Ring Expansions and O−H Insertions Zhang, W.; Romo, D. J. Org. Chem., 2007, 72, 8939-8942.
62. Simultaneous Arming and Structure/Activity Studies of Natural Products Employing O-H Insertions: An Expedient and Versatile Strategy for Natural Products-Based Chemical Genetics Peddibhotla, S.; Dang, Y.; Liu, J. O.; Romo, D. J. Am. Chem. Soc., 2007, 129, 12222-12231. [Highlighted in C&EN, October 1, 2007]
61. Substrate-Dependent Targeting of Eukaryotic Translation Initiation Factor 4A by Pateamine A: Negation of Domain-Linker Regulation of Activity Low, W.-K.; Dang, Y.; Bhat, S.; Romo, D.; Liu, J. O. Chem. & Biol., 2007, 14, 1-13.
60. Alkyl C-O Ring Cleavage of Bicyclic β-Lactones with Normant Reagents: Synthesis of Merck IND Intermediate Zhang, W.; Matla, A. S.; Romo, D. Org. Lett., 2007, 9, 2111-2114. [Highlighted in Organic Chemistry Portal: Organic Chemistry Highlights, December 10, 2007]
59. Concise Total Synthesis of (±)-Salinosporamide A, (±)-Cinnabaramide A, and Derivatives via a Bis-cyclization Process: Implications for a Biosynthetic Pathway? Ma, G.; Nguyen, H.; Romo, D. Org. Lett., 2007, 9, 2143-2146. [Highlighted in Synfacts 2007, 10, 1014]
58. Total Synthesis of (−)-Belactosin C and Derivatives via Double Diastereoselective Tandem Mukaiyama Aldol Lactonizations Cho, S. W.; Romo, D. Org. Lett., 2007, 9, 1537-1540. [Highlighted in Synfacts 2007, 8, 789]
57. Isolation and Identification of Eukaryotic Initiation Factor 4A as a Molecular Target for the Marine Natural Product Pateamine A Low, W.-K.; Dang, Y.; Schneider-Poetsch, T.; Shi, Z.; Choi, N. S.; Rzasa, R. M.; Shea, H. A.; Li, S.; Park, K.; Ma, G.; Romo, D.; Liu, J. O. Methods in Enzymology 2007, 431, 303-324.
2006
56. Eukaryotic Initiation Factor 2α-independent Pathway of Stress Granule Induction by the Natural Product Pateamine A Dang Y.; Kedersha, N.; Low, W.-K.; Romo, D.; Gorospe, M.; Kaufman, R.; Anderson, P.; Liu, J. O. J. Biol. Chem., 2006, 281, 32870-32878.
55. Synthesis, Characterization, and Utility of Thermoresponsive Natural/Unnatural Product Macroligands for Affinity Chromatography Zhou, M.; Sivaramakrishnan, A.; Ponnamperuma, K.; Low, W.-K.; Li, C.; Zhang, Y.; Liu, J. O.; Bergbreiter, D.; Romo, D. Org. Lett., 2006, 8, 5247-5250.
54. Total Synthesis and Comparative Analysis of Orlistat, Valilactone, and a Transposed Orlistat Derivative: Inhibitors of Fatty Acid Synthase Ma, G.; Zancanella, M.; Oyola, Y.; Richardson, R. D.; Smith, J. W.; Romo, D. Org. Lett., 2006, 8, 4497-4500.
53. Bicyclic- and Tricyclic-β-Lactones via an Organonucleophile Bis-cyclization of Keto-Acids: Enantioselective Synthesis of (+)-Dihydroplakevulin Henry-Riyad, H.; Lee, C. S.; Purohit, V.; Romo, D. Org. Lett., 2006, 8, 4363-4366.
52. Mild Deprotection of Primary N-(p-Toluenesulfonyl) Amides with SmI2 Following Trifluoroacetylation Moussa, Z.; Romo, D. Synlett 2006, 19, 3294-3298.
51. Diastereoselective, Vinylogous Mukaiyama Aldol Addition of Silyloxy Furans to Cyclic Ketones: Annulation of Butenolides and γ-Lactones Kong, K.; Romo, D. Org. Lett., 2006, 8, 2909-2912.
50. Planned and Unplanned Halogenations in Route to Selected Oroidin Alkaloids Wang, S.; Poullennec, K. G.; Dilley, A. S.; Romo, D. Tetrahedron 2006, 62, 7155-7161.
49. Practical, Catalytic, Asymmetric Synthesis of β-Lactones via a Sequential Ketene Dimerization/Hydrogenation Process: Inhibitors of the Thioesterase Domain of Fatty Acid Synthase Purohit, V. C.; Richardson, R. D., Smith, J. W.; Romo, D. J. Org. Chem., 2006, 71, 4549-4558.
48. A Unified Synthetic Stratgy Toward Oroidin Derived Alkaloids Premised on a Biosynthetic Proposal Dransfield, P. J.; Dilley, A. S.; Wang, S.; Romo, D. Tetrahedron 2006, 62, 5223-5247. [Bioinspired Approaches to Chemical Synthesis, invited paper]
2005
47. Inhibition of Eukaryotic Translation Initiation by the Marine Natural Product Pateamine A Low, W.-K.; Dang, Y.; Schneider-Poetsch, T.; Shi, Z; Choi, N. S.; Merrick, W. C.; Romo, D.; Liu, J. O. Mol. Cell 2005, 20, 709-722. [Highlighted in C&EN, December 19, 2005 and Featured in Chem. Biol. (Viewpoint by J. Clardy), 2006, 1, 17]
46. Synthesis, Structure, and Reactivity of Unexpectedly Stable Spiroepoxy-β-Lactones Obtained by Epoxidation of 4-Alkylidene-2-Oxetanones Duffy, R. J.; Morris, K. A.; Romo, D. J. Am. Chem. Soc., 2005, 127, 16754-16755.
45. Radical Reactions and α-Silylations of Optically Active 4-Trichloromethyl-β-Lactone Mitchell, A. T.; Romo, D. Heterocycles 2005, 66, 627-637. [Invited Paper]
44. Studies Toward a Marine Toxin Immunogen: Enantioselective Synthesis of the Spirocyclic Imine of (-)-Gymnodimine Kong, K.; Moussa, Z.; Romo, D. Org. Lett., 2005, 7, 5127-5130.
43. Asymmetric Synthesis of Bicyclic β-Lactones via the Intramolecular, Nucleophile-Catalyzed Aldol-lactonization (NCAL): Improved Efficiency and Expanded Scope Oh, S.; Cortez, G.; Romo D. J. Org. Chem., 2005, 70, 2835-2838. [Highlighted in Organic Chemistry Portal: Organic Chemistry Highlights, December 19, 2005]
42. Highly Regioselective Diels-Alder Reactions toward Oroidin Alkaloids: Use of a Tosylvinyl Moiety as a Nitrogen Masking Group with Adjustable Electronics Dransfield, P. J.; Wang, S.; Dilley, A.; Romo, D. Org. Lett., 2005, 7, 1679-1682.
2004
41. β-Lactones: Intermediates for Natural Product Total Synthesis and New Transformations Wang, Y.; Tennyson, R.; Romo, D. Heterocycles 2004, 64, 605-658.
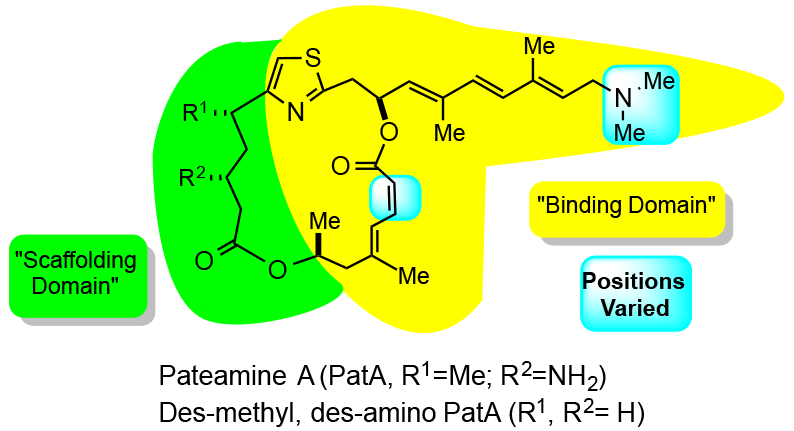
40. Evidence for Separate Binding and Scaffolding Domain in the Immunosuppressive and Antitumor Marine Natural Product, Pateamine A: Design, Synthesis, and Activity Studies Leading to a Potent, Simplified Derivative Romo, D.; Choi, N. S.; Buchler, I. P.; Shi, Z.; Liu, J. O. J. Am. Chem. Soc., 2004, 126, 10582-10588.
2003
39. Enantioselective Total Synthesis of (+)-Dibromophakellstatin Poullenec, K.; Romo, D. J. Am. Chem. Soc., 2003, 125, 6344-6345.
2002
38. Concise Total Synthesis of (+)-Brefeldin A: A Combined β-Lactone/Cross-Metathesis-Based Strategy Wang, Y.; Romo, D. Org. Lett., 2002, 4, 3231-3234.
37. Highly Diastereoselective Desymmetrizations of Cyclo(Pro,Pro): An Enantioselective Strategy Toward Phakellstatin and Phakellin Poullennec, K. G.; Kelly, A. T.; Romo, D. Org. Lett., 2002, 4, 2645-2648.
36. Nucleophilic Openings of Bicyclic β-Lactones via Acyl C-O and Alkyl C-O Cleavage: Catalytic, Asymmetric Synthesis of a Versatile, Carbocyclic Nucleoside Precursor and Protected Transpentenic Acid Yokota, Y.; Cortez, G. S.; Romo, D. Tetrahedron: Symposium in Print "Application of Strained Heterocycles as Reactive Intermediates in Organic Synthesis" Tetrahedron 2002 58, 7075-7080.
35. Mechanism and Origin of Stereoselectivity in Lewis Acid-Catalyzed [2+2] Cycloadditions of Ketenes with Aldehydes Singleton, D. A.; Wang, Y.; Yang, H. W.; Romo, D. Angew. Chem. Int. Ed., 2002, 41, 1572-1575.
34. A β-Lactone Route to Chiral γ-Substituted α-Amino Acids: Application to the Concise Synthesis of (S)- α-Azidobutyro Lactone and a Natural Amino Acid Tennyson, R.; Cortez, G. C.; Galicia, H. J.; Kreiman, C. R.; Thompson, C. M.; Romo, D. Org. Lett., 2002, 4, 533-536.
2001
33. Bicyclic β-Lactones via Intramolecluar NCAL Reactions with Cinchona Alkaloids: Effect of the C9-Substitutent on Enantioselectivity and Catalyst Conformation Cortez, G. S.; Oh S.-H.; Romo, D. Synthesis (Special Topics) 2001, 1731-1736.
32. Intramolecular Nucleophile Catalyzed Aldol-Lactonization (NCAL) Reactions: Catalytic, Asymmetric Synthesis of Bicyclic β-Lactones Cortez, G. S.; Tennyson, R.; Romo, D. J. Am. Chem. Soc., 2001, 123, 7945-7946. [Feature in Angew. Chem. Int. Ed. Highlights Article, Angew. Chem. Int. Ed., 2002, 41, 744-746]
31. Enantioselective Strategy to the Spirocyclic Core of Palau'amine and Related Bisguanidine Marine Alkaloids Dilley, A. S.; Romo, D. Org. Lett., 2001, 3, 1535-1538.
30. The Incredible Power of Organic Synthesis Romo, D. New Voices in Chemistry, Chemical and Engineering News-125th Anniversary Issue, March 26, 2001.
29. Studies Toward Gymnodimine: Development of a Single-Pot Hua Reaction for the Synthesis of Highly Hindered Cyclic Imines Ahn, Y.; Cardenas, G.; Yang, J.; Romo, D. Org. Lett., 2001, 3, 751-754.
2000
28. Use of In Situ Generated Ketene in the Wynberg β-Lactone Synthesis: New Transformations of the Dichlorinated β-Lactone Products Tennyson, R.; Romo, D. J. Org. Chem., 2000, 65, 7248-7252.
27. "Total Synthesis of Marine Natural Products Driven by Novel Structure, Potent Biological Activity, and/or Synthetic Methodology" In The Role of Natural Products in Drug Discovery, Romo, D.; Rzasa, R. M.; Schmitz, W. D.; Yang, J.; Cohn, S. T.; Buchler, I. P.; Shea, H. A.; Park, K.; Langehan, J. M.; Messerchmidt, N. B.; Cox, M. M., Mulzer, J.; Bohlmann, R., Eds.; Ernst Schering Research Foundation Workshop 32, Springer-Verlag Publishers, Berlin, Germany, 2000.
26. Studies Toward (-)-Gymnodimine: Concise Routes to the Tetrahydrofuran and Spirocyclic Moieties Yang, J.; Cohn, S.; Romo, D. Org. Lett., 2000, 2, 763-767.
1999
25. A Stereocomplementary Approach to β-Lactones: Highly Diastereoselective Synthesis of cis-β-Lactones, a β-Chloroacid, and a Tetrahydrofuran Wang, Y.; Zhao, C.; Romo, D. Org. Lett., 1999, 1, 1197-1199.
24. A Single Pot, Mild Conversion of β-Lactones to β-Lactams Yang, H. W.; Romo, D. J. Org. Chem., 1999, 64, 7657-7660.
23. Methods for the Synthesis of Optically Active β-Lactones (2-Oxetanones) Yang, H. W.; Romo, D. Tetrahedron 1999, 55, 6403-6434.
1998
22. Total Synthesis and Immunosuppressive Activity of (-)-Pateamine A and Related Compounds: Implementation of a β-Lactam-Based Macrocyclization Romo, D.; Rzasa, R. M.; Shea, H. A.; Park, K.; Langehan, J. M.; Sun, L.; Akhiezer, A.; Liu, J. O. J. Am. Chem. Soc., 1998, 120, 12237-12254.
21. Synthesis and Inhibitory Action on HMG-CoA Synthase of Racemic and Optically Active Oxetan-2-ones (β-Lactones) Romo, D.; Harrison, P. H. M.; Jenkins, S. I.; Riddoch, R. W.; Park, K. P.; Yang, H. W.; Zhao, C.; Wright, G. D. Bioorg. Med. Chem., 1998, 6, 1255-1272.
20. Simultaneous Deprotection and Purification of BOC-amines Based on Ionic Resin Capture Liu, Y.-S.; Zhao, C.; Bergbreiter, D. E.; Romo, D. J. Org. Chem., 1998, 63, 3471-3473.
19. Studies of the Asymmetric [2+2] Cycloaddition of Silylketenes and Aldehydes Employing Ti-TADDOL Catalysts Yang, H. W.; Romo, D. Tetrahedron Lett., 1998, 39, 2877-2880.
18. A β-Lactone-Based Strategy Applied to the Total Synthesis of (8S,21S,22S,23R)- and (8R,21S,22S,23R)-Okinonellin B Schmitz, W. D.; Messerschmidt, B.; Romo, D. J. Org. Chem., 1998, 63, 2058-2059.
17. Practical, One-Step Synthesis of Optically Active β-Lactones via the Tandem Mukaiyama Aldol-Lactonization (TMAL) Reaction Yang, H. W.; Romo, D. J. Org. Chem., 1998, 63, 1344-1347.
16. Total Synthesis of the Novel, Immunosuppressive Agent (−)-Pateamine A from Mycale sp. Employing a β-Lactam-Based Macrocyclization Rzasa, R. M.; Shea, H. A.; Romo, D. J. Am. Chem. Soc., 1998, 120, 591-592.
1997
15. A β-Lactone-Based Route to Cyclopentanes via Intramolecular Allylsilane Additions. An Unexpected Friedel-Crafts Alkylation Zhao, C.; Romo, D. Tetrahedron Lett., 1997, 38, 6537-6540.
14. Studies of the Tandem Mukaiyama Aldol-Lactonization (TMAL) Reaction: A Concise and Highly Diastereoselective Route to β-Lactones Applied to the Total Synthesis of the Potent Pancreatic Lipase Inhibitor, (−)-Panclicin D Yang, H. W.; Zhao, C.; Romo, D. Tetrahedron (Symposium in Print) 1997, 53, 16471-16488.
13. A Highly Diastereoselective, Tandem Mukaiyama Aldol-Lactonization Route to β-Lactones: Application to a Concise Synthesis of the Potent Pancreatic Lipase Inhibitor, (−)-Panclicin D Yang, H. W.; Romo, D. J. Org. Chem., 1997, 62, 4-5.
1996
12. A New Route to 2-Substituted Δ2-Thiazolines: Stille Cross-Couplings of 2-Bromo-Δ2-Thiazolines Schmitz, W. D.; Romo, D. Tetrahedron Lett. 1996, 37, 4857-4860.
11. Tandem Transacylation/Debenzylation of β-Lactones Mediated by FeCl3 Leading to γ- and δ-Lactones: Application to the Synthesis of (-)-Grandinolide Zemribo, R.; Champ, M. S.; Romo, D. Synlett 1996, 278-280.
1995
10. Highly Diastereoselective [2+2] Cycloadditions via Chelation Control: Asymmetric Synthesis of β-Lactones Zemribo, R. and Romo, D. Tetrahedron Lett., 1995, 36, 4159-4162.
9. Structural and Synthetic Studies of the Pateamines: Synthesis and Absolute Configuration of the Hydroxy Dienoate Fragment Rzasa, R. M.; Romo, D.; Stirling, D.; Blunt, J. W.; Munro, M. H. G. Tetrahedron Lett., 1995, 36, 5307-5310.
1994
8. Synthesis, Structure and Mechanism in Immunophilin Research Belshaw, P. J.; Meyer, S. D.; Johnson, D. D.; Romo, D.; Ikeda, Y.; Andrus, M.; Alberg, D. G.; Schultz, L. W.; Clardy, J.; Schreiber, S. L. Synlett 1994, 381-392.
7. Total Synthesis of (-)-Rapamycin Using an Evans-Tischenko Fragment Coupling Romo, D.; Meyer, S. D.; Johnson, D. D.; Schreiber, S. L. J. Am. Chem. Soc., 1993, 115, 7906-7907.
6. Synthetic Investigations of Rapamycin. 2. Synthesis of a C(22)-C(42) Fragment Romo, D.; Johnson, D. D.; Plamondon, L.; Miwa, T.; Schreiber, S. L. J. Org. Chem., 1992, 57, 5060-5063.
Graduate Publications
5. An Asymmetric Route to Enantiomerically Pure 1,2,3-Trisubstituted Cyclopropanes Romo, D.; Meyers, A. I. J. Org. Chem., 1992, 57, 6265-6270.
4. Chiral Non-Racemic Bicyclic Lactams. Vehicles for the Construction of Natural and Unnatural Products Containing Quaternary Carbon Centers Romo, D.; Meyers, A. I. Tetrahedron 1991, 47, 9503-9569.
3. Diastereoselective Cyclopropanations of Chiral Bicyclic Lactams Leading to Enantiomerically Pure Cyclopropanes. Application to the Total Synthesis of cis-(1S,3R)-Deltamethrinic acid and R-(-)-Dictyopterene C Romo, D.; Romine, J. L.; Midura, W.; Meyers, A. I. Tetrahedron 1990, 46, 4951-4994.
2. Chiral Bicyclic Lactams. Asymmetric Synthesis of cis-(1S,3R)-Deltamethrinic Acid Meyers, A. I.; Romo, D. Tetrahedron Lett., 1989, 30, 1745-1748.
Undergraduate Publications
1. Effects of Analogs of the Fungal Sexual Pheromone Sirenin on Male Gamete Motility in Allomyces Macrogynus Pommerville, J. C.; Strickland, J. B.; Romo, D.; Harding, K. E. Plant Physiology 1988, 88, 139-142.